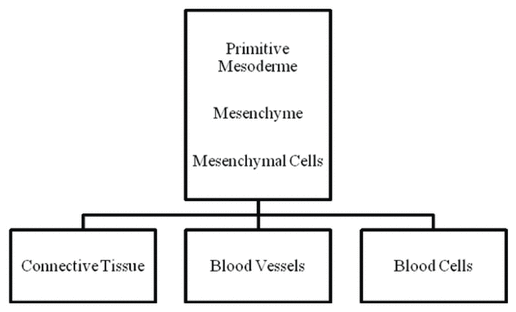
Figure 1 Mesenchymal cells are at the origin of connective, endothelial, and blood cells.
Bernard Payrau , MD *, Nadine Quéré , Physiotherapist , Danis Bois , PhD
CERAP, Fernando Pessoa University, Porto, Portugal.Background
A first study on vascular fasciatherapy enabled us to observe the turning of a turbulent blood flow into a laminar one, and a questioning on the process involved in this transformation emerged. The first question was: What is the nature of artery from the point of view of fascia? And a second question was: Which is the link permitting the observed process working in our first study? So this time, we are investigating a specific aspect of the big question that polarizes the interest of many researchers: “What is fascia?”
Methods
Following Donald Ingber’s statement, “It is necessary to understand how tissues and organs are structured across multiple size scales”, our research methods have been established in order to collect information on what is artery and what is fascia. Concerning these two organs, we have questioned science across the scales of embryology, anatomy, histology and cytology. Beyond the knowledge on structure, the functional link between artery and fascia is the necessary complement of this study whose starting point is in fact a questioning on process. As an application of this study, vascular fasciatherapy Danis-Bois Method and mechanotransduction have been investigated in theoretical and in research aspects to improve the understanding of how they work.
Results
The embryological approach points out a common origin and a histofunctional community of connective tissue and artery. As organs, arteries are sheathed by the adventia-fascia, and are penetrated by connective tissue extensions in media and intima. Furthermore, the functional point of view of this study reports the knowledge on mechanotransduction involving artery, both from the connective side and from the luminal side. Functional anatomy, surgery, histology, and cytology integrating the theory of the extended cytoskeleton, underline continuity from the static and functional points of view, with tensegrity being the architectural principle linking molecules to the entire body.
Conclusion
By answering these questions, we are attempting a better understanding of the mechanisms occurring in the progress of the arterial supporting point. One could presume that it relaxes adventitia and media, locally and all along the arterial network. Its action could also extend inward to the intima and on blood, as well as outwards to the neighboring connective tissue. By its local and remote action, it may be useful when diseases associate general perturbations and arterial disorders, like in high blood pressure or in aging.
KEYWORDS: fascias , fasciatherapy Danis Bois Method , arterial supporting point , embryology , histology , blood flow
After participation in the two first Fascia Research Congresses in Boston and in Amsterdam, we were preoccupied with the question, “What is fascia?” Definitions of fascia are so various that this underlines the obvious difficulty of answering this question; however, during these two congresses, several important answers emerged. Some insight came from a previous definition: “the dense irregular connective tissue that surrounds and connects every muscle, even the tiniest myofibril, and every single organ of the body, forming continuity throughout the body”(1,2). In the Amsterdam session, the continuity of the connective tissue as an integrating matrix of the body(3) was pointed out. This great interest of searchers has thrown a new light on the question of muscle and connective tissue. And in continuation of the study on blood flow in vascular fasciatherapy that we presented during the first research congress, a complementary orientation retained our interest in the question of what is fascia: arteries and connective tissue.
A first question arose about the nature of arteries from the point of view of fascia. Donald Ingber’s sentence guided our methods: “To understand how physical manipulation or movement of our body influence cellular biochemistry and tissue physiology, it is necessary to understand how tissues and organs are structured across multiple size scales”(4). So, we tried to understand the nature of artery and fascia by questioning science across the fields of embryology, anatomy, histology, and cytology. We first collected information from communications and publications issued by the two Fascia Research Congresses as pointing out the advanced research on the subject. Of course, a background of scientific papers published previously concerning fascia has been a basis of our research too. In embryological, cytological, histological, and anatomical domains, we also turned to a background of scholarly publications and to papers from scientific research from 1978 to 2009. Vascular research especially made a jump forward with the Nobel Prize awarded to Furchgott, Ignarro and Murad for having discovered the endogenous production of nitric oxide (NO). We relied on many publications issued since this period.
The second question was applied on the functional domain, by not considering any more fascia from the dissectional or topographical point of view, but with J. Van der Wal(3), with a more architectural or functional-mechanical understanding. Fasciatherapy section found a support in books and papers published mostly in French since 1984; the latest one was published in 2009. The research paper on vascular fasciatherapy was published in 2008 after presentation in the first Fascia Research Congress in 2007. The mechanotransduction section is based on various scientific papers published during a large period beginning in the ’90s, with many of them issued during the last five years. This question on the functional connections between fascia and artery which emerges throughout this research, opens a discussion based on hypothesizes coming from the beginning of the twentieth century and knowledge resulting from up-to-date research. Based on this, we propose a point of view on the static and functional connections between artery and fascia, and a better understanding of the mechanisms occurring in the progress of the arterial supporting point and its consequences on the body.
Connective tissue can be considered as central matrix of the body(3) and embryology is the first step for our questioning on the relationship between arteries and fascias. Two orientations will be followed: the origin of the connective tissue, and vasculogenesis.
Mesoderm is the middle layer of the three primary germ cell layers in the very early human embryo. It has been described as a germinal layer, an “inner tissue”(5), as opposed to the ectoderm and endoderm which are limiting tissues(3). It represents the matrix and the environment within which the organs and structures of the body have been differentiated and, therefore, are embedded(3).
The embryo’s mesenchyme is the basic tissue generated by the primitive mesoderm(6). It is the primary connective tissue of the body(3). The two main aspects of the nonspecialized connective tissue of the embryo are the mesenchymal tissue and the mucous connective tissue(6). Mesenchyme differentiates and generates every type of connective tissue and many organs in adults(3) including bone, muscle, and the middle layer of the skin, excepting nervous tissue and the digestive track(7).
Mesenchyme is organized in three components: cells, intercellular space, and fibers(5). Cells are undifferentiated and totipotent, and may be considered as one of the primary cells of development. They are at the origin of all the connective tissues, specialized or not, that can be observed after birth(6). In this study, one can notice that they are totally or partially at the origin of vascular endothelium and mesothelium (peritoneum, pleura, pericardium)(6). And this vascular endothelium is the origin of blood, which is also considered as specialized connective tissue(6).
As to functional development and differentiation of the mesenchyme, it is possible to consider two patterns referred to the principle of connection. These patterns are the two opposite applications: connection and disconnection. Connection generates the formation of a binding medium with fibers, interstitial substrate, and matrix, while disconnection generates the development of “intercellular space”(3). The lateral mesoderm splits into two layers and creates the inner or splanchnic layer that adheres to the endoderm, and the outer or somatic layer that becomes applied to the inner surface of the ectoderm.(7) The somatic layer completely lines the abdomino-thoracic cavity. The splanchnic layer, invaginating, delimits peritoneum, pleura, and pericardium, working so in the way of disconnection and creating space.
As bridges between somatic and splanchnic layers, when reflecting, the lateral mesoderm gives mesos that support and convey vessels and nerves to the thoraco-abdominal organs(7). Here appears the sheath role of the fascias for vessels (Figure 1).
|
|
||
|
Figure 1 Mesenchymal cells are at the origin of connective, endothelial, and blood cells. |
||
Vessels appearing during embryogenesis have been a subject of great interest for many studies, based on the avian model(8) on the embryo or on chicken-cell cultures. They have shown the mesoblastic origin of the vessels(9). In human development, around the embryo’s 18th day, the first cells of the vasculogenesis appear within the mesoderm(10) by the differentiation of totipotent undifferentiated mesoblastic cells into multipotent hemangioblasts.
Hemangioblasts have the double potency of differentiating into hematopoietic stem cells and into angioblasts according to the absence or the presence of the vascular endothelial growth factor (VEGF)(9). Angioblasts are the precursor of endothelial cells which associate in syncytium for constituting the primitive capillary plexus(9) within which the liquefaction causes the lumen to appear. Flattened angioblasts at the periphery form the endothelium(11), surrounded with extracellular matrix. Then, the primary vascular system constitutes by sprouting to form new capillaries, whereas primordial vascular smooth cells are recruited to the endothelium to form a multilayered vessel wall(12). Beyond this stage, vasculogenesis leads to the mature vascular system(10,13).
Neurovascular tract is one of the specified aspects of fascial tissue described as the extramuscular collagen fiber reinforcement of blood and lymph vessels, and of nerves(14). The function of fascial tissue is to sustain the vessels that are ramified upon it(15).
Leaving the traditional sheath definition of fascias, let’s adopt the architectural view of the connective tissue(3) showing the continuity of the structures. It is obviously an invitation to go on and penetrate organs and tissues towards the cells themselves and reach the intracellular intimacy.
Fascia interpenetrates and surrounds all organs(1). As surrounding vessels, fascias constitute tunic vessels, and are both connecting with and separating from the surrounding organs. As interpenetrating vessels, what are fascias within the arteries? Some answers can be found in histology(16).
Blood vessels are made of three layers consisting of three tunica. They are from the lumen: the intima, the media, and the adventitia. The tunica intima consists of an endothelium and any more or less subendothelial connective tissue. The tunica media is the layer of concentrically-arranged, smooth muscle cells and contains varying amounts of collagen fibers, elastic fibers, elastic lamellae, and proteoglycans secreted by the smooth muscle cells. The tunica adventitia is chiefly made of longitudinally arranged collagen fibers. So we can see that connective tissue can be found in the three layers of the arteries, with some differences according to their size. Actually, arteries are classified into three types according to their histology: large elastic arteries, medium muscular arteries (the majority of named arteries), and small arteries. These types are all continuous with one another but, according to type, the connective tissue differs.
Capillaries have the function of distributing blood in the body, bringing about an exchange between blood and tissues. Structurally, arteries carry and separate blood and tissues. The three-layered wall of the artery is connected to blood and to the surrounding tissues, and reacts so that it can be assimilated to a transducer. At a cytological scale, researcher’s interest focused on endothelial cells (ECs) and on vascular smooth muscle cells (VSMCs).
On the luminal side, ECs form the inner lining and constitute the ultimate interface between blood and artery, placing them as very important transducers. An extended surface layer, called the glycocalyx(17,18), is made up of several types of long proteoglycans anchored to the plasma membrane and seems to be an interposition between blood and endothelium.
Within the ECs cytoplasm is a cytoskeleton. Often compared to cellular scaffolding, it is an elaborate array of three types of protein filaments. The roles of the cytoskeleton are numerous: helping to maintain cell shape, producing locomotion, providing mechanical strength, modulating gene expression, aiding in chromosome separation during mitosis, and facilitating the intracellular transport of organelles(19). The cytoskeleton of the ECs physically connects the apical domain to the basal or lateral domains. It participates in cell-to-cell or cell-to-matrix junctions, and is essential to transduction because it is linking transmembrane proteins to cytoplasmic proteins(20).
Integrins go through the cell’s membrane, anchoring ECs to a basement membrane and linking connective tissue matrix fibers and the surrounding VSMCs and cytoskeleton(4). As a consequence, cytoskeleton and extracellular matrix are viewed as a single, synergetic, mechanically stabilized system, or the “extended cytoskeleton”(21). Thus, one can see a continuum from the inside of the cell towards the surface of the entire body, and from the point of view of the artery, the extended cytoskeleton can be seen as a connection between blood and skin.
Fascia research is a very wide domain and many studies are still to come. Some have already provided insight into this topic. Four issues attracted our interest concerning the question: “How is the link working between arteries and fascias?”
Fasciatherapy is a manual therapy(22,23,24), an alternative medicine founded by Danis Bois, PhD, Professor at the Fernando Pessoa University. He was first an osteopath and from these concepts and his personal discoveries, he developed a new approach to the fascias.
The specific touch of this method first evaluates the organization of the tensions and of the rhythms in the patient’s tissues. A slow spontaneous rhythmic movement called the Sensorial Movement in the Danis Bois Method (DBM) is flowing throughout every tissue in the entire body. This living organization of the body is a regulation force that supports vitality. When its dynamic presence in the tissues is felt through touch, it is a sign of a healthy body–mind with adaptability and self-healing potential. Additive tensions and perturbations of the rhythms in the body are the consequences of the tensed fascias in reaction to stress or disease. Fascia is a very sensitive tissue that detects any kind of stress — physical, emotional or psycho-social. It reacts by contracting and imprisoning the organs it covers, thus impairing their physiological functions. Furthermore, the tightening of their connective parts induces a perceptible disturbance in mobility and rhythm of these organs.
The therapeutic side of this touch is an application of gentle, superficial or deep pressures and stretching on the tissues, in accordance with the perception of the sensorial movement within the tissues. The supporting point that emerges at the end of this kind of stretching of the tissues is a stop of the therapeutic movement. Next occurs the phase of regulation of the patient’s body, resulting in a tangible reduction in muscle and tissue tone. By this means, restoring coherence and amplitude of the sensorial movement in the tissues is reached, inducing a release of tissue adherences, again of mobility, and a synchronization of the pulses which engage the body in its self-healing process. The remarkable point about the touching in this method is its very respectful approach which differentiates it from more aggressive techniques.
Pulsology is dedicated to the macro- and micro-circulation and to the blood, understood as a tissue. The pulse that can be perceived on the arteries is the result of transmission of the systolic blood impact on the aortic arch. So it is a vibratory complex running along the arterial network. The quality of this complex depends on many parameters. Two important ones are the tonic status of the arterial network and the quality of the tissues that they cross(30). For many centuries, the clinical semiology of the pulse has been a subject of great interest(31) and remains so now in our approach.
The arterial pulsology touch first evaluates the pulsologic status of the arterial network. Then, supporting points can be applied directly on the arteries. A supporting point is both an action and a perception by the hands of the practitioner. The action applies a special quality of pressure and the perception integrates the reactions to this pressure.
The nature of this pressure is both objective and subjective, being adaptive in feedback, according to the perception of the reactions of the artery and of the rest of the body.
Thus, the supporting point can be described as gentle (not to be received as an aggression), progressive (to be welcomed by the person in a pleasant depth), and enveloping (taking care not only of the artery but also of the entire body). The pressure must be uniform, global, and respectfully adapted to the outlines of the zone to be treated(25).
The purpose of this pressure is to create a reflexlocalized vasodilation, managing a real blood tide towards this zone(25). A good duration is determined by the time during which the modifications of the pulse are still developing. This quality of pulse analysis has a double interest: to be curative by its pumping action, and to be informing on the degree of liberation of the tissues(25).
The feeling of a vasculodynamic event modulating the arterial tonus emerges during this short period of some seconds(32).
The reaction of the artery is not just a vasodilation. The practitioner has the perception of the arterial reaction as a tone modulation. Actually, it is less a quantitative modification of the pulse (a frequency change), than a qualitative modulation. The modifications of the arterial tone at the supporting point induce a vibratory change perceived by the practitioner. Normalization of the pulse is reached when the shock wave becomes slow, soft, flexible, generous and diffusing, extending into the surrounding tissues where immediate consequences develop(25).
The extending wave of normalization takes two directions: first, in spreading along the arterial network according to the arterio-arterial reflex; second, in diffusing to the surrounding connective tissues. This extension can be evaluated with precision by the practitioner who perceives and evaluates the local reaction which then becomes regional, and finally general, developing a chain of fascia reactions throughout the body.
We evaluate the time for getting this reaction, just as we evaluate the remaining asymmetries and loss of synchronization of the pulses(25). Normalization of the pulses is obtained when this time is short, the pulses are synchronous, and tensions and adhesions are overcome(25) (Figure 2).
|
|
||
|
Figure 2 The supporting point. |
||
Applying fasciatherapy DBM with a pulsology touch allows one to modify the type of the arterial blood flow(33), turning a turbulent flow into a laminar one. Sixteen patients’ responses to a short application of a standardized vascular fasciatherapy treatment versus a standardized conventional body massage were recorded on Echo Doppler tracings. Clinostatism was the marker in both treatments. The vascular fasciatherapeutic treatment had the following protocol: 10 minutes treatment including a time of treatment of the abdominal aorta and of the femoral and popliteal arteries using the technique of the supporting point. The tracings were realized on the femoral artery, before and after resting and after treatment. The result was a harmonization of blood velocities on the traces by the increase of the dark windows considered as reflecting the decrease of turbulences (Figure 3).
|
|
||
|
Figure 3 Dark windows increase considered as reflecting the decrease of turbulences. |
||
A study on arteriolar dilation in response to skeletal muscle contraction(34) provides the evidence that a matricryptic, heparin-binding site within fibronectin fibrils of adult connective tissue plays a dynamic role in regulating both vascular responses and vascular tone. Tensile forces change the conformation of fibronectin fibers surrounding the arteriolar wall. It seems that skeletal muscle contraction alters the conformation of extracellular matrice (ECM) fibronectin surrounding skeletal and/or smooth muscle cells. The first step in these reactions could be the tension applied to the fibrils initiated by the muscular contraction. By the means of mechanotransduction, tensile forces are translated into a biochemical process leading to a release of nitric oxide by the smooth vessel cells or by the skeletal muscle itself, thereby producing a vasodilation. No implication of neurovegetative regulation is enrolled in this mechanism.
Furthermore, according to the degree of tension, ECM fibronectin fibrils play a dynamic role in regulating arteriolar response during exercise and vascular tone while resting. On the contrary, chronic low-level muscle contraction may cause a blunting or down-regulation of this pathway. This study allows us to hypothesize that a sufficient level of traction applied on the connective tissue may obtain a good trophicity and adaptability.
Mechanotransduction covers all the molecular mechanisms by which cells sense mechanical forces and convert them into changes in intracellular biochemistry and gene expression(4). Mechanotransduction has been and still is the center of many studies concerning arteries. These studies have mainly been applied to the “blood face” of the arteries, because forces associated with blood flow are major determinants of vascular morphogenesis and physiology(18) and, obviously, of atherogenesis.
Two main types of forces are applied by blood flow on the luminal side of the arteries: shear stress and blood pressure. The first one acts on the ECs on a longitudinal way, whereas blood pressure exerts a circumferential stretch normal to the vessel wall on both the ECs and the VSMCs.
Responding to mechanical forces, both ECs and VSMCs modulate artery diameters so that the blood flow meets the demands of the tissues(18). The mechanotransduction-induced EC adaptive processes represent a case of the “Wisdom of the Cell”, as a part of the more general concept of the “Wisdom of the Body” promulgated by Cannon, to maintain cellular homeostasis in the face of external perturbations(35). ECs respond to increased blood flow by causing relaxation of the surrounding VSMCs. VSMC relaxation in response to flow occurs over seconds to minutes and if high flow persists, remodeling of the artery wall enlarges the lumen over time in a period of weeks to months(36). Decreased flow induces vessel narrowing(37), and extreme low flow may lead to complete vessel regression, which involves apoptosis of the ECs(38).
Most of the responses to flow are the fact of ECs, which indicate that ECs must express specific mechanotransducers(18). Many hypothetic mechanotransducers have been proposed to function in sensing flow, but many questions still need answers. Let’s list the main components:
Originally, the role attributed to theses fibrils was to be signal conductors(39). Cytoskeleton of the ECs physically connects different regions of the ECs, the apical domain where shear stress is applied, to the basal or lateral domains where high strain and mechanotransduction events have been observed(40). This suggests transmission of forces (force flow) to these sites(41) through the cytoskeleton. But there is little evidence that the cytoskeleton functions as a direct mechanotransducer of shear stress per se(18).
Some lateral integrins may stimulate endothelial nitric oxide (NO) synthase, contributing to NO release. The same process is observed for the basal surface cell integrins that bind to the ECM beneath the ECs. Mechanotransduction involves integrin receptors and occurs at cell matrix adhesion sites(42) and may have a role in vasorelaxation. However, there is little evidence to suggest that integrins function as direct mechanotranducers in the shear stress response(18).
Elements on the luminal side of the EC that directly experience shear stress may mediate flow response: mechanosensitive ion channels (a quick response potassium channel has been identified), primary cilia(18), glycocalyx. Glycocalyx is a layer of several hundred microns decorating the surface of ECs with a wide variety of membrane-bound macromolecules(17). It is an element of a functional endothelium with “the evidence of the surface glycocalyx acting as a mechanotransducer”(43). Due to its situation and thickness, shear stress should not be exerted directly on the plasma membrane but must be transmitted through the glycocalyx(44). Fluid shear stress is transmitted to the cell through the core proteins of the glycocalyx, and the specific connections of these proteins to the actin cytoskeleton (syndecans) and the plasma membrane (glypicans) mediate specific cell signaling (e.g. NO production, cytoskeletal reorganization)(43). However, whereas the role of the glycocalyx in flow-sensing is intriguing, its exact contribution remains uncertain(18).
Another way for mechanical forces to be applied on the arteries is the connective tissue surrounding them, but this has been the focus of less interest. Thus, the study on skeletal muscle contraction inducing local vasodilation is innovating, and points out a novel mechanotransduction pathway for transmitting mechanical forces from the ECM to cells(34). Other studies on this are expected.
The lowering of turbulences under an arterial supporting point implies that a physical change occurred in the arteries and/or in the blood by the means of the fasciatherapeutic treatment. So what is fascia tissue in artery, and what is changing during and after the supporting point?
Connective tissue is issued from the mesoderm layer in the embryo. Many organs originate from the undifferentiated mesodermic cells, just like fascias and arteries. They differentiate into hemangioblasts, from which appear endothelium and blood cells. Unity, as hypothesized by Jordan in 1916 (cited by Corvol(9)), can also be noticed here. Endothelium and blood cells’ common mesodermic precursor cell origin makes a great difference with nerves that are sheathed by fascias, but come from the embryo’s ectoderm layer.
According to its binding function, mesoderm produces the adventitia sheathing arteries with extensions connecting the vessel to the surrounding connective tissue. Each tunic of artery contains more or less connective tissue interpenetrating media and intima as prolongation of adventitia.
Mesoderm, by its spacing function, creates the lumen of artery providing room for blood movement, as it does with pericardium, pleura or peritoneum for heart, lungs or bowels. They are lined up and delimitated by an epithelium called endothelium in vessels, while in body cavities it is named mesothelium.
We think that because of their origin, connective tissue and organs originating in the same source can keep a histofunctional community in humans for the span of their lives.
Functional anatomy enlightens the continuity of the connective tissue as an integrating matrix of the body(3). Strolling under the skin with J.C. Guimberteau, one can see “this network known as connective tissue, the binding material of our living matter(45)”.
The human body seems to be made of the only and same tissue which is functionally differentiated: there are only tissue connections, simply a histological continuum without any clear separation between the skin and hypodermis, the vessels, the aponeurosis, and the muscles(46). So connective tissue, its cells, MEC, and fibers are an obvious link in this construction.
On a smaller scale, the cell unit also participates in this unity. The extended cytoskeleton concept(21) introduces the link between biologic modulations and macroscopic modifications. The cytoskeleton and the extracellular matrix being viewed as a single, synergetic, mechanically stabilized system lets us explain how movement is transmitted to tissues and cells by being translated into a biochemical process(21). Although information transmission was usually considered to be the role of the nervous system and blood circulation, connective tissue appears to be also on duty as another pathway. And this network transmits information in both ways, from inside to outside the cells and to the more superficial layers of the body and vice versa. When considering the artery, this type of relationship lets us think that a mechanical event on the skin may affect arteries and blood, whereas modification of blood or ECs may also have repercussion towards superficial tissues. So, in arteries this mechanotransduction process may lead to a release of NO by the smooth vessel cells from stimulation, either from distant tissues or from the lumen.
The theory of tensegrity emerged from the interests of architects (from Richard Buckminster Fuller to Rene Motro) and biologists (Donald Ingber(47)), and their meeting point of connection with our discussion can be found in these definitions: “a type of prestressed structural network, composed of opposing tension and compression elements that self-stabilizes its shape through establishment of a mechanical force balance”, and “tensegrity is used to stabilize the shape of living cells, tissue and organs, as well as our whole bodies”(4). Hence, the use of this architectural system for structural organization provides a mechanism to physically integrate part and whole(4).
Functionally, transmission of tension through a tensegrity array provides a means to distribute forces to all interconnected elements and, at the same time, to couple, or “tune,” the whole system mechanically as one.(47). This may look like clever disorganization: “Although chaotic in appearance, this interplay between vacuoles and fibrils creates mechanical stability between the forces of local compression and overall tension. The result equation is one of equilibrium. And in this organization, arteries look perfectly adapted to the connective tissue moves”(45).
Tensegrity at the cellular level has allowed us to comprehend better how cellular shape and mechanical forces — such as pressure in blood vessels or compression in bone —influence the activities of genes(47).
The existence of a local arterial regulation not taking into account neurovegetative action is well known. The mechanisms of arterial tonus and motility regulation respond to several pathways. Some are dependant of blood flow quality variations. Contraction of the small arteries by myogenic effects from stretching the artery is the kind of mechanism working in response to acute changes of blood pressure(30). Shear stress controls vascular physiology in adults by NO release (also prostacyclin, arachidonic metabolites, potassium ions), within ECs when blood flow increases. Some other reactions seem to be induced by modifications of the surroundings of the artery: vasodilation by NO release from tensing the ECM fibrils(34) that could be a novel mechanotransduction pathway. It may be just an example of a common approach to transmitting mechanical forces, including pulsatile flow and shear stress, from the ECM to cells(34).
Arteries have a special relation with fascias. Connective tissue is present in the three tunics of the artery. Adventitia is a typical sheathing fascia, which becomes tense in reaction to stress. Media is an association of muscle and connective tissue reacting to local mechanical variations (i.e. blood pressure) or general influence (i.e. stress) by tensing and/or by contracting. Intima, whose endothelium can be assimilated to a very big autocrine/paracrine formation(48) reacting mainly to the influence of blood qualities (i.e. type of flow, components), lies on a connective layer underlining endothelium.
Because of their blood transporting role, arteries have a very specific situation, being the interface between the liquid tissue (blood) that is the most interior part of the body, and the solid tissue that is connected to the most exterior part of the body. Furthermore, according to the anatomic and functional unity concept, arteries are much more than transporters; they are one of the main actors of the body.
The vascular fasciatherapeutic touch engages the artery reaction which relaxes adventitia and media, and regulates its tone and motricity locally and along the arterial network. This action is both local and diffusing to the connective tissue and the endothelium.
Given the nature of the flow being under the dependence of the pressure and the intrinsic qualities of the vessel in which the blood passes, the arterial physical change produces a decrease of turbulent flow and, thereby, an increase of laminar flow. The gain in laminar flow modulates shear stress and stimulates a nitric oxide (NO) release, inducing arterial relaxation and anti-inflammatory response in endothelium(33), potentially reducing the development of atherosclerosis (as can be observed on carotid bifurcation, a classical place of turbulences).
Consequence on blood qualities are not the only effect of this touch. A diffusion of effects also occurs in the surrounding tissues which diffuse by the means of the extended cytoskeleton and tensegrity(33).
In situ effects, developing in distant and global expression, can be seen from different points of view. The fasciatherapeutic practitioner explains that he perceives the internal movement producing under his hands vasodilation and the diffusion of the effects as a continuum through the entire body. The biological point of view is the tension-relaxation process of fascia, with its extension to the cytoskeleton at a microscopic scale and, at a macroscopic scale, to the full development of the fascia network via the extended cytoskeleton on the tensegrity mode.
Perturbed arterial vasomotion is obviously a domain of preference of this manual therapy. Although looking like a relaxation technique, the supporting point also implies fibril stretching. Maintaining a sufficient level of traction applied on the connective tissue permits keeping or restoring a good trophicity and adaptability in vasoregulation(34).
As treatment of an arterial spot diffuses so easily along the arterial network and through the entire body, vascular fasciatherapy may be useful when diseases associate general perturbations and arterial disorders. When psychosocial stress and dysfunction of the vascular/cardiovascular system are linked, as in high blood pressure(49) or in aging(34), vascular fasciatherapy seems to be a good response. By its action on the vasodynamic aspects of endothelial Payrau : SUPPORTING POINT APPLIED ON ARTERIES dysfunction(33), vascular fasciatherapy may also be a therapy for chronic inflammation(50).
Many studies on many fronts remain to be conducted in this field of research. Vascular fasciatherapy works on homeostatic regulation locally and in the whole body. Many questions still remain about how homeostasis operates, and they are as difficult to answer as the questions on “wisdom of the body”, and “wisdom of the cell”(35). Some elements of response have emerged, in particular with the increased knowledge on the role of cytoskeleton, whereby some people have hypothesized that it is participating in a conscious state(39).
For further research, the following areas should be of interest: interaction between fascia and arteries, consequences of the touch on vasomotion and endothelial dysfunction, study of the action of vascular fasciatherapy on stress, HBP and inflammatory diseases. By carrying out this research, we attempted to become better acquainted with our results, and to discover the mechanisms involved in reaching our goals.
1
Schleip R. Fascial plasticity – a new neurobiological explanation: part 2.
J Bodyw Mov Ther
. 2003;7(2):104–116.
2
Schleip R. Fascial plasticity – a new neurobiological explanation: part 1.
J Bodyw Mov Ther
. 2003;7(1):11–19.
3 Van der Wal JC. The architecture of the connective tissue in the musculoskeletal system – an often overlooked functional parameter as to proprioception in the locomotor apparatus. In: Fascia Research II, proceedings of the Second Fascia Congress. Ulm University, The Netherlands: FRC; 2009. p. 21–35.
4
Ingber DE. Tensegrity and mechanotransduction.
J Bodyw Mov Ther
. 2008;12(3):198–200.

5
Blechschmidt E. Anatomie und ontogenese des menschen. Biologische Arbeitsbücher Vol 22. Heidelberg, Germany: Quelle und Meyer; 1978.

6
Balas D and Philip P. Cours d’histologie générale. http://homepage.mac.com/danielbalas/HISTOLOGIE/HISTGENE/histgen1/histgen2/histgen2.html. Accessed February 3, 2010.

7
Goffinet M. Cours d’embryologie médicale. University of Louvain, Medical School, Brussels, Belgium. http://www.md.ucl.ac.be/didac/anat110/Squelette.html. Accessed December 12, 2008.

8
Dieterlen-Lievre F, Jaffredot T, Pardanaud L. L’émergence du réseau endothélial pendant la vie embryonnaire. Pathologie et biologie. 1999;47(4):301–306.
9 Corvol MP. Modelage et remodelage vasculaire, 2001. http://www.college-de-france.fr/media/med_exp/UPL25223_pcorvol.pdf. Accessed December 12, 2008.
10 Philippe HJ. Embryologie du coeur. http://www.heartandcoeur.com/definition/formation_coeur.php. Accessed December 12, 2008.
11
Sabin FR. Preliminary note on the differentiation of angioblasts and the method by which they produce blood-vessels, blood-plasma and red blood-cells as seen in the living chick.
The Anatomical Record
. 2005;13(4):199–204.
12
Hungerford IE and Little CD. Developmental biology of the vascular smooth muscle cell: building a multilayered vessel wall.
J Vasc Res
. 1999;36(1):2–27.

13
Favier J. Angiogenese normale et pathologique: mecanismes cellulaires et moléculaires, cibles pharmacologiques. http://www.dea-cardiopneumo.org/doc_telechargement/biblio_c/2006_2007/BCP2/Favier.pdf. Accessed December 12, 2008.

14 First International Fascia Research Congress. Glossary of terms. http://www.fascia2007.com/glossary.htm. Accessed December 2008.
15
Langevin HM and Huijing PA. Communicating about fascia: history, pitfalls, and recommendations.
IJTMB
. 2009;2(4):3–8. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3091474/. Accessed December 12, 2009.
16 University of Ottawa, Faculty of Medicine. Histology of the blood vessels. http://www.courseweb.uottawa.ca/medicine-histology/English/Cardiovascular/HistologyBloodVessels.htm. Accessed December 15, 2009.
17
Pries AR, Secomb TW, Gaehtgens P. The endothelial surface layer.
Pflugers Arch Eur J Physiol
. 2000;440(5):653–666.
18
Hahn C and Schwartz MA. Mechanotransduction in vascular physiology and atherogenesis.
Nat Rev Mol Cell Biol
. 2009;10(1):53–62.


19 Cytoskeleton Research. What is the cytoskeleton? http://www.cytoskeletons.com/. Accessed December 15, 2009.
20
Doherty GJ and Mc Mahon HT. Mediation, modulation and consequences of membrane-cytoskeleton interactions.
Ann Rev Biophys
. 2008;37:65–95.
21
Ingber DE. Cellular tensegrity: defining new rules of biological design that govern the cytoskeleton.
J Cell Sci
. 1993;104:613–627.
22 Bois D. The wild region of lived experience. Berkeley, CA: North Atlantic Books; 2009.
23 Courraud C. Fasciathérapie et sport: le match de la santé. Paris. France, Point d’Appui; 1999.
24
Eschalier I. La fasciathérapie, une nouvelle méthode pour le bien-être. Paris, France: Le Cherche Midi; 2005.

25
Bois D. Concepts fondamentaux de fasciathérapie et de pulsologie profonde. Paris, France: Editions Maloine; 1984.

26 Bois D. Fascias, sang, rythmes, complices dans les pathologies fonctionnelles. Paris, France: SPEK; 1985.
27 Bois D. La vie entre les mains. Paris. France: Trédaniel; 1989.
28
Bois D, Berger E, Alessandri J, et al. Une thérapie manuelle de la profondeur. Paris, France: Trédaniel; 1990.

29 Quere N. La pulsologie, action sur les fascias, le système vasculaire et la microcirculation. Paris, France: Point d’Appui; 2004.
30
Payrau B. Le pouls dans tous ses états. Available from: http://bibliosource.free.fr.
31
Diderot D, d’Alembert J. Encyclopédie, ou dictionnaire raisonné des sciences, des arts et des métiers. Available from: http://encyclopedie.uchicago.edu/.

32 Quéré N. Pulsology in fasciatherapy-somatology DBM [DVD]. Paris, France: Point d’Appui; 2007.
33
Quéré N, Noel E, Lieutaud A, d’Alessio P. Fasciatherapy combined with pulsology touch induces changes in blood turbulence potentially beneficial for vascular endothelium.
J Bodyw Mov Ther
. 2008;13(3):239–245.
34
Hocking DC, Titus PA, Sumagrin R, Sarelius IH. Extracellular matrix fibronectin mechanically couples skeletal muscle contraction with local vasodilation.
Circ Res
. 2008;102(3):372–379.
35
Chien S. Mechanotransduction and endothelial cell homeostasis: the wisdom of the cell.
AJP-Heart Circ Physiol
. 2007;292(3):1209–1224.
36
Di Stefano I, Koopmans DR, Langille BL. Modulation of arterial growth of the rabbit carotid artery associated with experimental elevation of blood flow.
J Vasc Res
. 1998;35(1):1–7.

37
Browlee RD and Langille BL. Arterial adaptations to altered blood flow.
Can J Physiol Pharmacol
. 1991;69(7):978–983.
38
Baffert F, Le T, Sennino B, et al. Cellular changes in normal blood capillaries undergoing regression after inhibition of VEGF signaling.
AJP-Heart Circ Physiol
. 2006;290(2): 547–559.
39
Frixione E. Recurring views on the structure and function of the cytoskeleton: a 300-year epic.
Cell Motility and the Cytoskeleton
. 2000;46(2):73–94.

40
Davies FP. Flow-mediated endothelial mechanotransduction.
Physiol Rev
. 1995;75(3):519–560.
41
Helmke BP, Thakker DB, Goldman RD, Davies PF. Spatiotemporal analysis of flow-induced intermediate filaments displacement in living endothelial cells.
Biophys J
. 2001;80(1): 184–194.

42
Chen CS, Tan J, Tien J. Mechanotransduction at cell-matrix and cell-cell contacts.
Ann Rev Biomed Eng
. 2004;6:275–302.
43
Tarbell JM and Pahakis MY. Mechanotransduction and the glycocalyx.
J Intern Med
. 2006;259:339–350.

44
Weinbaum S, Tarbell JM, Damiano ER. The structure and function of the endothelial glycocalyx layer.
Ann Rev Biomed Eng
. 2007;9:121–167.
45 Guimberteau JC. Strolling under the skin. Paris, France: Elsevier; 2004.
46
Guimberteau JC, Sentucq-Rigall J, Panconi B, et al. Introduction à la connaissance du glissement des structures souscutanées humaines.
Annales de Chirurgie Plastique Esthétique
. 2005,50(1):19–36.
47
Ingber D. The architecture of life.
Sci Am
. 1998;278(1):48–57.
48
Besson N. Analyse biochimique du bis (monoacylglycérol) phosphate dans les cellules monocytes THP-1 et étude de son rôle sur le trafic intracellulaire du cholestérol [Thèse, Spécialité biochimie]. Lyon, France: Institut national des Sciences Appliquées de Lyon; 2006.

49 Payrau B. Can fasciatherapy combined with pulsology touch improve blood pressure regulation of HBP patients? [Post-grad dissertation]. Porto, Portugal: Fernando Pessoa University; 2009.
50
Quéré N. La fasciathérapie méthode Danis Bois et les fascias sous l’éclairage des recherches scientifiques actuelles. La fasciathérapie et ses impacts tissulaires, vasculaires, cellulaires et biochimiques [Masters dissertation]. Porto, Portugal: Fernando Pessoa University; 2009.

COPYRIGHT
Published under the CreativeCommons Attribution NonCommercial-NoDerivs 3.0 License.
CONFLICT OF INTEREST NOTIFICATION
The author declares that there are no conflicts of interest.
INTERNATIONAL JOURNAL OF THERAPEUTIC MASSAGE AND BODYWORK , VOLUME 4 , NUMBER 4 , December 2011