Danielle Gentile, PhD1*, Danielle Boselli, MS2, Susan Yaguda, MSN,RN1, Rebecca Greiner, PhD, PA-C1, Chase Bailey-Dorton, MD, MSPH1
1Department of Supportive Oncology, Levine Cancer Institute, Atrium Health, Charlotte, NC, USA
2Department of Cancer Biostatistics, Levine Cancer Institute, Atrium Health, Charlotte, NC, USA
Background
Healing Touch (HT) and Oncology Massage (OM) are nonpharmacologic pain interventions, yet a comparative effectiveness study has not been conducted for pain in breast cancer.
Purpose
This breast cancer subgroup analysis compared the effectiveness of HT vs. OM on pain.
Setting
The research occurred at an outpatient setting at an academic hybrid, multi-site, community-based cancer institute and Department of Supportive Oncology across four regional locations.
Participants
Breast cancer outpatients along the cancer continuum who experienced routine clinical, nonexperimentally manipulated HT or OM.
Research Design
The study was an observational, retrospective, comparative effectiveness post hoc subanalysis of a larger dataset. Patients reporting pain < 2 were excluded. Pre- and posttherapy pain scores and differences were calculated. Logistic regression modeled posttherapy pain by modality, adjusting for pretherapy pain. The proportions experiencing ≥ 2-point (clinically significant) pain reduction were compared with chi-square tests.
Intervention
The study focused on the first session of either HT or OM.
Main Outcome Measures
Pre- and posttherapy pain (range: 0 = no pain to 10 = worst possible pain).
Results
A total of 407 patients reported pre- and posttherapy pain scores, comprised of 233 (57.3%) who received HT and 174 (42.8%) who received OM. Pretherapy mean pain was higher in HT (M=5.1, ± 2.3) than OM (M=4.3, ± 2.1) (p < .001); posttherapy mean pain remained higher in HT (M=2.7, ± 2.2) than OM (M=1.9, ± 1.7) (p < .001). Mean difference in pain reduction was 2.4 for both HT and OM. Both HT (p < .001) and OM (p < .001) were associated with reduced pain. Proportions of clinically significant pain reduction were similar (65.7% HT and 69.0% OM, p = .483). Modality was not associated with pain improvement (p = .072).
Conclusions
Both HT and OM were associated with clinically significant pain improvement. Future research should explore attitudes toward the modalities and potential influence of cancer stage and treatment status on modality self-selection.
KEYWORDS: breast cancer, cancer pain, healing touch, oncology massage, nonpharmacologic pain management
Breast cancer is the most commonly diagnosed cancer in women, and the second leading cause of cancer deaths among women.(1) Although rare, breast cancer is not limited to cisgender women, and also appears in other gender identities including cisgender men and transgender populations.(2) Approximately one-third of patients report breast pain before breast cancer surgery,(3) and persistent pain after surgery endures for between 60%(4) and 66% of patients.(3,5) Such pain is associated with psychological distress, physical disability, and impaired daily life activities,(6) all decreasing quality of life. The National Comprehensive Cancer Network’s (NCCN) pain management guidelines emphasize physical, cognitive, and spiritual nonpharmacologic pain management tools.(7) The American Society of Clinical Oncology (ASCO) pain management guidelines also emphasize the importance of nonpharmacologic integrative therapies.(8) Furthermore, The Joint Commission requires nonpharmacologic pain treatment modalities for hospital accreditation.(9) Healing Touch and Oncology Massage are used to manage cancer-related pain and are endorsed for supportive care specifically for those with breast cancer by ASCO and experts within the Integrative Oncology field.(10,11)
Based on ancient Eastern healing practices, HT is a biofield therapy energy medicine practice that aims to restore and balance energy that has been disrupted due to stress, illness, injury, grief, various medical procedures, and cancer treatments such as chemotherapy, surgery, radiation, and hormone therapy.(12) Energy medicine involves manipulating the body’s naturally occurring energy fields to channel the body’s innate healing abilities.(13) Energy healing is the “channeling of healing energy through the hands of a practitioner into the client’s body to restore a normal energy balance and, therefore, health.”(14) HT practitioners may use light, gentle touch, sweeping hand motions with their hands near the body or both to restore and balance energy interrupted by physical and emotional stressors.(15) It has demonstrated ability to improve health-related quality-of-life and reduce respiratory rate, heart rate, blood pressure, pain, mood disturbances, and fatigue.(12) In a randomized controlled trial on women with cancer, HT produced better health-related quality of life, physical functioning, and vitality with reduced pain.(16) Multiple studies suggest HT reduces cancer-related pain,(17,18) including in those with breast cancer,(19) but further studies are needed.(12)
OM differs from other types of massage in that it is practiced by massage therapists specifically trained in techniques for cancer patients’ unique needs, with attention to the cancer treatment journey and disease process.(20) OM can help to ameliorate quality-of-life issues associated with cancer and its treatments. Techniques are customized by adjusting the treatment applications of positioning, pressure, pace, and/or site to accommodate medical devices, side-effects of drug treatments, and discomfort or pain associated with cancer and its treatments.(21) A subgroup meta-analysis of massage on breast cancer patients’ pain revealed significant effects of reduced pain in multiple studies,(22) although further randomized controlled trials on the effect of massage on breast cancer pain are needed.(23) Quality of life and coping abilities are also strengthened after OM.(24) A systematic review of OM randomized control studies suggests that massage relieves pain, nausea, stress, depression, anxiety, and fatigue, while improving sleep and mental clarity. However, additional high methodological quality studies are warranted.(25)
Comparative effectiveness research (CER) provides evidence for patients and clinicians to make informed decisions about which methods of health-care delivery may be most beneficial.(26,27) CER can be of particular use when studying general clinical practice with a diversity of healthcare delivery features which may not translate to the results of tightly controlled randomized clinical trials. Essentially, CER can illuminate potential differences in the effectiveness of therapies in “real world” settings.(28) CER studies may be conducted using observational, retrospective data as is the case within the current research.(29) The current comparison of HT to OM is important due to the similarities between the modalities. Patients may self-select to modality, and it would be clinically useful to determine if there is evidence for one being more helpful for pain than the other. If this is the case, clinicians may steer patients toward the modality with better efficacy.
Few studies compare HT to OM for pain improvement. Post-White and colleagues(30) found that pain was significantly reduced by both HT and OM, although HT was not directly compared to massage. A study of outcomes after reiki (an energy therapy related to HT), massage, and yoga found no significant differences across the modalities in pain reduction.(31) These studies were all conducted with fewer than 165 participants. Gentile and colleagues(32) compared HT to OM for pain management in a general sample of 572 cancer patients and found both HT and OM provided immediate clinically significant pain relief after a single therapy session. After adjusting for pretherapy pain scores, OM was found to be associated with increased odds of pain improvement. Within this sample, 50.8% of those accessing HT and OM were breast cancer patients, making more detailed examination of this population particularly salient. While previous research suggests that both HT and OM are promising integrative therapies, sample sizes in most studies have been small and, to our knowledge, none involve directly comparing the effectiveness of HT and OM in a breast cancer patient population.
It is particularly important to study the comparative effectiveness of HT vs. OM on pain within the breast cancer population because breast cancer patients are mainly cisgender women who are known to be higher utilizers of complementary and integrative therapies.(33) Furthermore, within our Integrative Oncology clinic, breast cancer patients comprise the majority of complementary and integrative modality users. Exploring the comparative effectiveness of different modalities on pain may help us to provide targeted recommendations for limited resources that are in demand by patients of all cancer types, thereby maximizing efficiency.
Given the frequent use of HT and OM by breast cancer patients, and the lack of research comparing the effectiveness of HT to OM on pain in breast cancer patients, the purpose of this post hoc subset analysis of a larger study was to answer:
Do HT and/or OM reduce pain scores (measured from 0 = no pain to 10 = worst possible pain)?
Do HT and/or OM provide clinically significant pain improvement (defined as ≥ 2-point reduction)?(34)
Are the proportions of clinically significant pain improvement significantly different across HT and OM in patients with severe pretherapy pain (defined as ≥ 7 pain score)?(35)
The methodological design was an observational, pretest/posttest, retrospective analysis of a single therapy session of either HT or OM. This report was a breast cancer only subpopulation analysis of a previous, larger study that included all cancer types.(32)
Participants were breast cancer outpatients along all phases of the cancer continuum (recently diagnosed, in treatment, long-term survivorship). There were no inclusion or exclusion criteria as this is a retrospective analysis of all patients who received routine therapy. Both therapies (HT, OM) occurred in an outpatient setting at an academic hybrid, multisite, community-based cancer institute within a Department of Supportive Oncology.
This observational, retrospective study included a cohesive, consecutive sample of all patients who received HT or OM. The analytic sample was created by applying exclusions described in the Data Collection and Management section below.
Patients were either referred to receive therapy by a health-care provider or self-referred. As a verbal process leading to referrals, clinicians’ descriptions of both HT and OM were unstandardized; explanations were tailored to individuals’ needs and questions. Patients were able to self-select the therapy modality they preferred. HT and OM practitioners provided routine, clinical care therapy for patients across all aspects of the cancer continuum (recently diagnosed, in treatment, long-term survivorship)(36) for approximately 45 minutes, and documented therapy techniques in the electronic medical record.
Healing Touch techniques were tailored to individual needs. Patients received HT individually in a private room with soft lighting and calming music. Patients remained clothed and lay in a supine position on a padded table. Bolsters were available to support positioning to patients’ preferences. They could be covered by a sheet, if desired. Prior to administering HT, the practitioner and patient set an intention for the patient’s highest good. Then the practitioner achieved a state of homeostasis in the patient’s human energy field using various techniques. Light touch or near-body touch techniques included: 1) Magnetic Clearing to clear congestion from the Human Energy Field which is defined as a “luminous field of energy that comprises a person, extends beyond the physical body, and is in a continuous mutual process with the environmental energy field. It is a vital energy that is a continuous whole and is recognized by its unique pattern; it is dynamic, creative, nonlinear, unpredictable, and flows in lower and higher frequencies. The balanced HEF is characterized by flow, rhythm, symmetry, and gentle vibration”(37); 2) Mind Clearing to decrease stress and promote relaxation; and 3) The Chakra Connection to connect, open, and balance the energy centers (chakras). The HT therapist adhered to a Standard Operating Procedure which called for a comprehensive medical review of each patient at every visit. All HT sessions were covered by philanthropic funds and provided by a single HT therapist.
Oncology Massage utilized gliding, rhythmic strokes, gentle kneading, and gentle tissue holds to meet individual therapeutic goals. Before therapy, medical conditions were assessed to determine technique modifications. Modifications included alternative positioning (e.g., side lying or seated vs. traditional prone/supine positioning) and additional cushioning to reduce pressure on wounds, tumors, medical devices, and surgical sites. Private, dedicated rooms were used for individual massage sessions. The rooms had soft lighting, calming music, and a space heater and fan to adjust for patient comfort. Hypoallergenic, unscented lotion was used as lubricant. Pressure on the Walton Pressure Scale occurred on levels 1–3, with 3 typically being reserved for those who have completed their cancer treatments.(38) OM therapists also adhered to a Standard Operating Procedure and reviewed the patient’s medical history at each visit. Four OM therapists provided OM across four regional locations. Patients were often treated by the same therapist for return visits. OM was offered as a self-pay service at $45 per visit, which is more affordable than within most community settings.(39) Reimbursement forms were provided to those patients whose health-care insurers covered OM. To reduce cost barriers, scholarships from philanthropic funds to cover visit costs were available.
Description of therapists’ training and scope of practice is considered an important variable in studies which focuses on interventions.(40) OM therapists had 600–900 base hours of training in Therapeutic Massage and Bodywork and 25–48 hours of OM-specific training. OM training was received from Tracy Walton & Associates (Tracey Walton Muscular Therapy, Cambridge, MA) and Oncology Massage Workshop with Vickie Torrey (Costa Rica School of Massage Therapy). At the beginning of data collection, the range of years of experience among the four OM therapists was 7 to 36 yrs. The HT practitioner was certified as a Level 6 practitioner and instructor, which is the highest recognized certification possible. The HT practitioner had 15 years of experience at the beginning of data collection. Both HT and OM therapists completed yearly continuing education and performance review through the Levine Cancer Institute (Charlotte, NC).
From January 2015 to March 2019, a cohesive, consecutive sample of all patients given therapy self-evaluated their pain immediately before and after receiving a single therapy session of HT or OM on a scale from 0 = no pain to 10 = worst possible pain. This scale is similar to the validated Edmonton Symptom Inventory (ESAS-r scale)(41) and permitted as an informal adaptation by the ESAS-r authors. The data were exported from an internal, prospective, growing database that is managed by clinic staff in efforts of ongoing quality improvement initiatives and research. Data were managed in REDCap, a secure, web-based, electronic data capture tool.(42) The institutional review board of Atrium Health approved this retrospective analysis with consent waiver (IRB File # 10-17-17E) before data analysis. There were 1,504 HT and 1,213 OM therapy administrations captured during the data collection period, of which 1,491 HT and 1,208 OM records were complete (i.e., included pre- and posttherapy pain scores and therapy date). To avoid confounding therapy effects, the data were restricted to the first therapy visit per patient regardless of modality (322 HT records, 256 OM records remaining). Those reporting pretherapy pain <2 were excluded as they could not achieve clinically significant pain improvement (defined as a pain score decrease of at least 2 points on the 0 to 10 scale).(34) Approximately 30% of observations (27.6% HT, 32.0% OM) were excluded due to pretherapy pain <2. The final analytic sample was 407; 57.3% (n = 233) HT and 42.8% (n = 174) OM observations.
This report sought to establish and compare the efficacies of HT and OM therapies in achieving clinically significant pain improvement in breast cancer patients. Demographics and history of anticancer treatments were summarized by modality, alongside summary statistics of pretherapy and posttherapy pain scores and paired differences. Assessment of select characteristics was performed using chi-square tests; analysis of pain scores and paired differences were performed using two-sample and paired t tests, respectively. The primary objective was evaluated using logistic regression models, estimating the odds of clinically significant pain improvement by modality adjusted for pretherapy pain score, age, and anticancer treatment type (i.e., surgery, chemotherapy, radiation, hormone therapy). Backwards elimination was used in model selection (significance level < .05). The model-adjusted proportions of pain improvement were estimated through the multivariate model at the mean value of pretherapy pain. A subgroup analysis of pain improvement proportions in those with severe pain ( ≥ 7 pretherapy pain(35,41,43)) was performed. Statistical analysis was performed using SAS, version 14.1 (SAS Institute, Inc., Cary, NC).
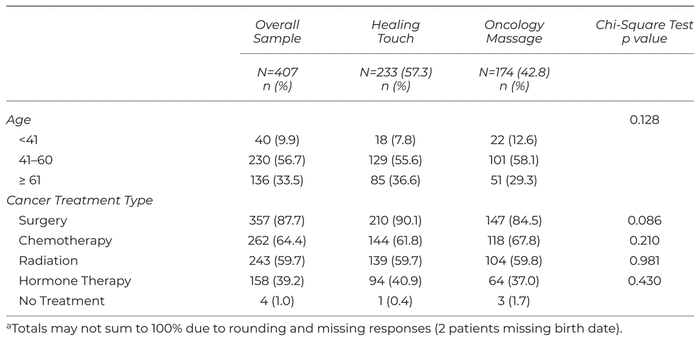
Patients were mostly treated with chemotherapy (64.4%) or surgery (87.7%). Cancer treatment histories were similar between the modalities (Table 1). Pretherapy pain score among all patients ranged from 2 to 10, with an average of 4.7. HT patients presented with higher pretherapy pain (HT χ̄ =5.1 ± 2.3 vs. OM χ̄ =4.3 ± 2.1; p < .001) and also reported higher post-therapy pain (HT χ̄ = 2.7 ± 2.2 vs. OM χ̄ = 1.9 ± 1.7; p < .001) than OM. Both HT and OM patients reported statistically significant reductions in numerical pain score (HT χ̄ = −2.4, p < .001; OM χ̄ = −2.4, p < .001).
Table 1 Patient and Treatment Characteristics (n = 407)a
Immediate pain improvement of a reduction in pain score of at least one point on the 10-point scale was observed in 88.7% of all patients. Less than one percent reported (n= 3) a higher pain score after therapy.
Over sixty-seven percent (n = 273) reported immediate clinically significant pain improvement (i.e., reduction in pain score of ≥ 2 points); proportions were similar between the modalities (HT 65.7% vs. OM 69%; p = .483). In a final multivariate model, pretherapy pain score for both groups taken into account for the comparison (OR = 1.41, 95% CI (1.26, 1.59); p < .001) was associated with increased odds of clinically significant pain improvement, but modality was not (OM vs. HT: OR = 1.51, 95% CI (0.96, 2.36); p = .072). The difference in model-predicted probabilities of pain improvement (HT = 70%, OM = 74%) was similar to that between the unadjusted proportions.
The previous analyses were re-estimated in a subset of 97 (23.8%) patients with severe pretherapy pain only (i.e., pain score of 7–10); 29.2% of HT and 16.7% of OM presented with severe pretherapy pain (p = .003). The proportions of clinically significant pain improvement were 80.9% in HT and 89.7% in OM (p = .287). Neither baseline pain (OR = 0.67, 95% CI (0.39, 1.14); p = .142) nor modality (OM vs. HT: OR = 2.03, 95% CI (0.53, 7.85); p = .305) were associated with pain improvement.
The purpose was to compare the effectiveness of HT vs. OM on pain in breast cancer patients receiving HT and OM. To our knowledge, this report is the first to examine and compare the two integrative modalities focusing on pain in a breast cancer population. Results suggested that both 69% of OM and 65.7% of HT patients reported significant pain improvement after one therapy session. Independent of pretherapy pain score, OM was associated with increased odds of pain improvement. Those who received HT had higher mean pretherapy pain than OM, and also reported experiencing higher pain scores after therapy than OM. In those with severe pretherapy pain, there were no statistically significant differences in clinically significant pain improvement. We speculate that those in greater pain may self-select HT over OM. They may perceive HT’s light touch and touch-free modifications as more comfortable than their perception of OM as more direct muscle manipulation. Financial costs and physicians’ descriptions of each modality may also have influenced self-selection of either HT or OM.
The results of the current breast cancer-only subpopulation resembled the larger, all-cancer-types comparison of HT to OM for pain outcomes by Gentile and colleagues.(32) In both the breast cancer only subset and the larger heterogenous cancer type dataset, there was no evidence for one modality having a greater efficacy in pain reduction over the other. The reductions in mean pain score were also similar. HT and OM both had a reduction of 2.4 points on a 0 to 10 scale within the breast cancer subpopulation, and in the larger heterogeneous dataset had reductions of 2.4 for HT and 2.5 for OM. This is likely associated with the breast cancer subtype making up the largest proportion (50.8%) of the all-cancer-type sample. While the results were similar, the extant study reports the first comparison of HT to OM within the breast cancer population. This is an important contribution to the literature given that the breast cancer subgroup tends to be high users of integrative and complementary modalities.(33) These findings bolster previous research suggesting that both(30,32) HT(12,16) and massage(17,18,44,45) effectively reduce cancer-related pain. However, it is difficult to compare directly the effect sizes of the current study to the extant literature due to differences in study populations and pain outcome measures. Further study is needed; yet these findings suggest HT and OM are both helpful nonpharmacologic therapies for pain improvement.
This report achieves valuable scientific insights, yet it has limitations. The etiology of pain (i.e., directly related to cancer or not) and pain mechanism (e.g., neuropathic, nociceptive) was unassessed, and patients’ reports of pain were subject to self-perception. Pain improvement was observed was from a pre- to posttherapy timeframe of approximately 45 minutes, and it is unknown if pain relief continued and for how long. Highly experienced, licensed, and credentialed practitioners provided HT and OM. Therefore, results may not generalize to other HT and OM therapy settings with practitioners who have different credentials and levels of experience. Therapy experiences differed across sessions because each treatment was customized to fit individual patient goals. The observational, retrospective design did not allow randomization to therapy modality, and was unable to account for underlying unknown variables in self-selection between the therapies. The difference in HT being entirely covered by philanthropy while OM was self-pay with scholarships available may have been a factor in differences across HT and OM selection. This study was also unable to control for potential confounding characteristics and medical history (e.g., cancer stage, use of OTC analgesics and/or opioids, expectations for pain relief) because these variables were not collected as part of the brief in-clinic assessment, and manual data abstraction from the medical record for a large number of cases was infeasible. The study was also limited in focus to cisgender female breast cancer patients, as consistent with the majority of extant breast cancer research. However, the results focused on a breast cancer population in a nonexperimentally manipulated, routine clinical environment. This design improves generalizability to other oncology care settings.
This report raises important inquiries for further study, including exploration of breast cancer patients’ attitudes toward HT compared to OM and how attitudes may differ with different levels of pretherapy pain, cancer treatment, and other factors. Insight on attitudes may determine influence on self-selection to therapy modality. Study of HT and OM on pain in community settings outside of cancer institutions would be beneficial. Additionally, future research should explore how long pain improvement across both modalities endures, and the optimal number of treatment sessions for lasting pain relief. The impact of HT and OM on pain in breast cancer stratified by cancer stage would be valuable. For many breast cancer survivors, joint pain is a common side-effect of endocrine therapies, which can lead patients to discontinue potentially life-prolonging therapy.(46) It would be interesting to study if HT or OM could reduce this specific type of pain in this subset of breast cancer survivors to potentially improve endocrine therapy adherence. Finally, research should explore how HT and OM impact pain in cancer patients taking opioids and other pharmacologic treatments such as non-steroidal anti-inflammatory drugs. It would be interesting to explore if adding HT and/or OM to pharmacologic pain management therapies may help reduce drug intake.
The study of pain management in breast cancer is enhanced through this report’s comparison of HT to OM for pain response after a single therapy session in a sample of breast cancer patients. Both modalities provided clinically significant pain relief, which provides empirical evidence for the use of either modality in those with breast cancer experiencing pain. HT and OM meet NCCN, ASCO, and The Joint Commission guidelines for nonpharmacologic pain management, and may be useful options for those who wish to use integrative therapies to ameliorate pain. This project demonstrates the feasibility of collecting pre-and posttherapy symptoms during routine clinical practice which can later be studied with retrospective analysis.
Pain is a frequently occurring, stressful side-effect of cancer and its associated treatments. This report compared the immediate effectiveness of HT and OM on pain in a sample of breast cancer patients after one routine clinical session. Both HT and OM were associated with significantly reduced pain score and clinically significant pain improvement, although the duration of the pain relief was unknown. Both OM and HT were associated with clinically significant pain improvement when controlling for pretherapy pain. In severe pretherapy pain, the odds of clinically significant pain improvement between HT and OM were not significantly different. These findings represent noteworthy contributions to the study of each modality’s effects by demonstrating that both HT & OM provided immediate, clinically significant pain relief after a single therapy session in a routine clinical breast cancer population. Future research should examine the potential influence of patients’ attitudes toward each modality and cancer treatment and stage on self-selection of either HT or OM. These integrative therapies meet NCCN and ASCO guidelines for nonpharmacologic pain management.(7,8)
The authors gratefully acknowledge the 24 Foundation and Sherry Strong Foundation which provided funds to provide Healing Touch and Oncology Massage services. This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
The authors declare there are no conflicts of interest.
1 National Breast Cancer Foundation Inc. Breast Cancer Facts. Dallas, TX: the Foundation; 2016. Retrieved from: https://www.nationalbreastcancer.org/breast-cancer-facts
2 de Blok CJM, Wiepjes CM, Nota NM, van Engelen K, Adank MA, Dreijerink KMA, et al. Breast cancer risk in transgender people receiving hormone treatment: nationwide cohort study in the Netherlands. BMJ. 2019:l1652.


3 Langford DJ, Schmidt B, Levine JD, Abrams G, Elboim C, Esserman L, et al. Preoperative breast pain predicts persistent breast pain and disability after breast cancer surgery. J Pain Sympt Manage. 2015;49(6):981–994.
4 Wang L, Guyatt GH, Kennedy SA, Romerosa B, Kwon HY, Kaushal A, et al. Predictors of persistent pain after breast cancer surgery: a systematic review and meta-analysis of observational studies. Can Med Assoc J. 2016;188(14):E352–E361.
5 Poleshuck EL, Katz J, Andrus CH, Hogan LA, Jung BF, Kulick DI, et al. Risk factors for chronic pain following breast cancer surgery: a prospective study. J Pain. 2006;7(9):626–634.


6 Chiang DLC, Rice DA, Helsby NA, Somogyi AA, Kluger MT. The prevalence, impact, and risk factors for persistent pain after breast cancer surgery in a New Zealand population. Pain Med. 2019;20(9):1803–1814.

7 Swarm RA, Abernethy AP, Anghelescu DL, Benedetti C, Buga S, Cleeland C, et al. Adult cancer pain. J Natl Compr Cancer Netw. 2013;11(8):992–1022.
8 Paice JA, Portenoy R, Lacchetti C, Campbell T, Cheville A, Citron M, et al. Management of chronic pain in survivors of adult cancers: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016;34(27):3325–3345.

9 The Joint Commission. Standards Revisions Related to Pain Assessment and Management. Oakbrook Terrace, IL: TJC; 2017. Available from: https://www.jointcommission.org/assets/1/18/HAP_Pain_Jan2018_Prepub.pdf
10 Greenlee H, DuPont-Reyes MJ, Balneaves LG, Carlson LE, Cohen MR, Deng G, et al. Clinical practice guidelines on the evidence-based use of integrative therapies during and after breast cancer treatment. CA. 2017;67(3):194–232.

11 Lyman GH, Greenlee H, Bohlke K, Bao T, Demichele AM, Deng GE, et al. Integrative therapies during and after breast cancer treatment: ASCO endorsement of the SIO Clinical Practice Guideline. J Clin Oncol. 2018;36(25):2647–2655.

12 Anderson JG, Taylor AG. Effects of healing touch in clinical practice: a systematic review of randomized clinical trials. J Holistic Nurs. 2011;29(3):221–228.
13 National Center for Complementary and Integrative Health. Terms Related to Complementary and Integrative Health: energy healing therapy. Bethesda, MD: The Canter; 2020. Retrieved from: https://www.nccih.nih.gov/health/providers/terms-related-to-complementary-and-integrative-health
14 Rao A, Hickman LD, Sibbritt D, Newton PJ, Phillips JL. Is energy healing an effective non-pharmacological therapy for improving symptom management of chronic illnesses? A systematic review. Complem Ther Clin Pract. 2016;25:26–41.
15 Agdal R, von B Hjelmborg J, Johannessen H. Energy healing for cancer: a critical review. Forschende Komplementarmedizin. 2011;18(3):146–154.

16 Cook CA, Guerrerio JF, Slater VE. Healing touch and quality of life in women receiving radiation treatment for cancer: a randomized controlled trial. Alt Ther Health Med. 2004;10(3):34–41.
17 Celebiŏglu A, Gürol A, Yildirim ZK, Büyükavci M. Effects of massage therapy on pain and anxiety arising from intrathecal therapy or bone marrow aspiration in children with cancer. Int J Nurs Pract. 2015;21(6):797–804.
18 Jane SW, Liao MN, Lee SH, Lin YC. The effects of a massage intervention in patients with cancer pain: an evidence-based approach [in Mandarin]. Hu Li Za Zhi. 2014;61(6):23–28.
19 Guy J-B, Bard-Reboul S, Trone J-C, Vallard A, Espenel S, Langrand-Escure J, et al. Healing touch in radiation therapy: is the benefit tangible? Oncotarget. 2017;8(46):81485.


20 Society for Oncology Massage. What is Oncology Massage? [FAQ website page]. Richmond, MN: The Society; 2020. Retrieved from: https://www.s4om.org/clients-and-patients/clients-patients-faq#:~:text=An%20oncology%20massage%20is%20a,of%20chemotherapy%2C%20radiation%20and%20surgery
21 Sood A, Barton DL, Bauer BA, Loprinzi CL. A critical review of complementary therapies for cancer-related fatigue. Integr Cancer Ther. 2007;6(1):8–13.

22 Lee S-H, Kim J-Y, Yeo S, Kim S-H, Lim S. Meta-analysis of massage therapy on cancer pain. Integr Cancer Ther. 2015;14(4):297–304.

23 Lee MS, Lee EN, Ernst E. Massage therapy for breast cancer patients: a systematic review. Ann Oncol. 2011;22(6):1459–1461.

24 Sagar SM, Dryden T, Wong RK. Massage therapy for cancer patients: a reciprocal relationship between body and mind. Curr Oncol. 2007;14(2):45–56.


25 Ernst E. Massage therapy for cancer palliation and supportive care: a systematic review of randomised clinical trials. Support Care Cancer. 2009;17(4): 333–337.

26 Sox HC. Comparative effectiveness research: a report from the Institute of Medicine. Ann Intern Med. 2009;151(3):203–205.

27 Iglehart JK. Prioritizing comparative-effectiveness research—IOM recommendations. New Eng J Med. 2009;361(4):325–328.

28 Velentgas P, Dreyer NA, Nourjah P, Smith SR, Torchia MM, eds. Developing a Protocol for Observational Comparative Effectiveness Research: a User’s Guide. Rockville, MD: Agency for Healthcare Research and Quality; 2013.
29 Berger ML, Mamdani M, Atkins D, Johnson ML. Good research practices for comparative effectiveness research: defining, reporting and interpreting nonrandomized studies of treatment effects using secondary data sources: The ISPOR Good Research Practices for Retrospective Database Analysis Task Force—Part 1. Value Health. 2009;12(8):1044–1052.

30 Post-White J, Kinney ME, Savik K, Gau JB, Wilcox C, Lerner I. Therapeutic massage and healing touch improve symptoms in cancer. Integr Cancer Ther. 2003;2(4):332–344.
31 Rosenbaum MS, von de Velde J. The effects of yoga, massage, and reiki on patient well-being at a cancer resource center. Clin J Oncol Nurs. 2016;20(3):E77–E81.

32 Gentile D, Boselli D, O’Neill G, Yaguda S, Bailey-Dorton C, Eaton TA. Cancer pain relief after healing touch and massage. J Altern Complem Med. 2018;24(9–10):968–973.
33 Bishop FL, Lewith GT. Who uses CAM? A narrative review of demographic characteristics and health factors associated with CAM use. Evid-based Complem Altern Med. 2010;7(1):11–28.
34 Younger J, McCue R, Mackey S. Pain outcomes: a brief review of instruments and techniques. Curr Pain Headache Rep. 2009;13(1):39–43.


35 Serlin RC, Mendoza TR, Nakamura Y, Edwards KR, Cleeland CS. When is cancer pain mild, moderate or severe? Grading pain severity by its interference with function. Pain. 1995;61(2):277–284.

36 Hopkins J, Mumber MP. Patient navigation through the cancer care continuum: an overview. J Oncol Pract. 2009;5(4):150–152.


37 Shields D, Fuller A, Resnicoff M, Butcher HK, Frisch N. Human energy field: a concept analysis. J Holistic Nurs. 2017;35(4):352–368.
38 Walton T. The massage therapy pressure scale. In: Walton T. Medical Conditions and Massage Therapy: a Decision Tree Approach. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2011.
39 American Massage Therapy Association. Massage Therapy Industry Fact Sheet. Evanston, IL: The Association; 2018. Retrieved from: https://www.amtamassage.org/infocenter/economic_industry-fact-sheet.html
40 Boutron I, Altman DG, Moher D, Schulz KF, Ravaud P. CONSORT statement for randomized trials of nonpharmacologic treatments: a 2017 update and a CONSORT Extension for Nonpharmacologic Trial Abstracts. Ann Intern Med. 2017;167(1):40–47.

41 Hui D, Bruera E. The Edmonton Symptom Assessment System 25 years later: past, present, and future developments. J Pain Sympt Manage. 2017;53(3):630–643.
42 Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381.

43 Oldenmenger WH, Pleun J, de Klerk C, van der Rijt CC. Cut points on 0–10 numeric rating scales for symptoms included in the Edmonton Symptom Assessment Scale in cancer patients: a systematic review. J Pain Symptom Manage. 2013;45(6): 1083–1093.
44 Dion LJ, Engen DJ, Lemaine V, Lawson DK, Brock CG, Thomley BS, et al. Massage therapy alone and in combination with meditation for breast cancer patients undergoing autologous tissue reconstruction: a randomized pilot study. Complem Ther Clin Pract. 2016;23:82–87.
45 Collinge W, MacDonald G, Walton T. Massage in supportive cancer care. Semin Oncol Nurs. 2012;28(1):45–54.

46 Hershman DL, Unger JM, Greenlee H, Capodice JL, Lew DL, Darke AK, et al. Effect of acupuncture vs. sham acupuncture or waitlist control on joint pain related to aromatase inhibitors among women with early-stage breast cancer: a randomized clinical trial. JAMA. 2018;320(2):167–176.


Published under the CreativeCommons Attribution-NonCommercial-NoDerivs 3.0 License. ( Return to Text )
INTERNATIONAL JOURNAL OF THERAPEUTIC MASSAGE AND BODYWORK, VOLUME 14, NUMBER 1, March 2021