Katharina L. Wiest , PhD, MSPH 1 * , Victoria J. Asphaug , MScPH 1 , Kathryn E. Carr , MPH 1 , Emily A. Gowen , LMT 2 , Timothy T. Hartnett , MSW, MHA 1
1 Research Division, CODA, Inc., Portland, OR, USA
2 4423 NE Tillamook St., Portland, OR, USA
Background:
Chronic pain is a common cause of health care utilization and high levels of pain are pronounced in individuals engaged in methadone maintenance treatment. Although massage has been demonstrated to alleviate chronic pain symptoms, its use as an adjunctive therapy to modify pain during opioid-replacement treatment is absent from the literature.
Purpose:
To consider the efficacy of Swedish massage in reducing pain in opioid-dependent patients with chronic pain receiving methadone treatment.
Setting:
Trial was conducted at a nonprofit methadone treatment center serving low-income patients.
Research Design:
A randomized clinical trial with randomized to either 1) massage plus treatment-as-usual (TAU) (n = 27) or 2) TAU (n = 24). Durability of treatment effect was evaluated at Week 12.
Intervention:
Eight weekly 50-minute Swedish massage sessions plus TAU or TAU alone.
Main Outcome Measures:
Pain, anxiety, depression, physical functioning, decreased substance use, and improvement in treatment engagement.
Results:
Randomized participants were comparable at Baseline for demographic, pain, physical, and emotional variables. Massage group reported improved pain scores; worst pain had a clinically significant 2-point improvement while the other pain scores did not. Overall improvements were not observed in treatment engagement or levels of anxiety, depression, or physical functioning. A subgroup of the participants, who felt they could be pain-free, consistently reported improvements in pain from Baseline to Week 8, and this was most pronounced and clinically significant in the massage group.
Conclusions:
These preliminary findings do not support an overall clinically significant positive effect of Swedish massage on reduction in pain ratings or improvement in anxiety, depression, or treatment engagement in a substance-using, opioid-dependent population with chronic pain. Future nonpharmacologic pain research in marginalized substance-using populations may wish to consider some of the challenges and limitations faced in this project.
KEYWORDS: massage , pain , opioid dependence , substance use , self-efficacy , methadone
Chronic pain is a common cause of health care utilization and represents a major health concern.(1) The overlap of chronic pain and substance use has been recognized since early in the twentieth century(2) and has been examined extensively with a focus on chronic pain in patients engaged in care.(3–14) Relatively high self-reports of pain are of particular concern within substance-use treatment populations, and are most pronounced for individuals engaged in methadone maintenance treatment.(4,15–17) Estimates of chronic pain in opioid-dependent patients receiving methadone ranges from 37% to 80%.(4) In sharp contrast, the estimated chronic pain prevalence in the general population ranges from 11%(18) to 30.7%.(1)
Since the 1960s, methadone has been used successfully for treatment of opioid dependence.(19) For patients with chronic pain prior to starting methadone treatment, over half reported that their pain predated their addiction(20) and 44% identified pain as their reason for enrolling in methadone treatment rather than other opioid-dependence treatment options.(16) Chronic pain patients had more extensive addiction treatment histories, more severe opioid cravings, earlier age at first opioid use, and more psychiatric and health concerns.(13) Individuals with dependence on opioid analgesics reported higher levels of chronic pain(16) and stated that their pain contributed to their addiction.(3) Patients with chronic pain had higher average daily methadone doses,(6) more severe patterns of polysubstance use, and more medical and social problems.(7) Chronic pain was associated with negative treatment outcomes, such as medical and psychiatric comorbidity and counselor frustration.(9) In this population, nonpharmacologic options for chronic pain management are limited.
The term “massage therapy” encompasses many different touch therapy techniques. Swedish massage is the most widely studied and reported massage technique in the literature.(21) Swedish massage incorporates moderate pressure touching(22) and has minimal risk of adverse events, making it appropriate for protocol standardization and comparisons across studies. Using a standardized Swedish massage protocol, clinical research in healthy adults found a cumulative positive biologic effect which varied with massage frequency.(22,23) While the precise biologic mechanism through which massage effects pain is unclear,(24) consistent support exists for the beneficial effects for treating chronic pain, especially chronic low-back pain.(21) A 2007 review of clinical trials found massage was superior to relaxation therapy, acupuncture, and self-help education.(21,25) Complementary and Alternative Medicine (CAM) therapies are viewed as highly effective in substance-use treatment populations(26) and specifically in opioid-dependent patients.(9)
Randomized controlled trial research on use of massage as an adjunctive therapy to modify pain during opioid-replacement treatment is lacking. The goal of this pilot project was to begin to initiate consideration of massage’s potential as an adjunctive nonpharmacologic pain treatment in substance-using, opioid-dependent patients receiving methadone maintenance.
In this 12-week randomized clinical trial (RCT), participants were assigned to either an intervention arm of eight weekly 50-minute Swedish massage sessions and treatment-as-usual (TAU) (n = 27) or to TAU alone (n = 24), with a follow-up visit four weeks after completing the intervention. Sample size was determined by minimum number needed to detect a 20% difference between groups. The study was approved by a local academic institutional review board and participants provided written informed consent. Participants randomized to the massage arm completed an additional massage informed consent.
Study participants were recruited from a large, nonprofit medication-assisted treatment program for opioid dependence. Eligibility criteria required that participants be age 18 or older, meet DSM-IV-TR(27) criteria for opioid dependence, receive methadone treatment for opioid dependence for at least 90 days, report nonneuropathic chronic pain at intake of intensity ≥ 4 on a 0 to 10 numeric rating scale (NRS),(28) be willing to forgo any nonstudy-related massage treatment, remain in methadone therapy for the study duration, and provide informed consent. Exclusionary conditions, determined by the medical staff in conjunction with the lead licensed massage therapist (LMT), included: pregnancy; pending incarceration; severe and persistent mental health concerns;(29) broken bones, fractures, dislocations, or severe sprains; open or unhealed sores or wounds such as injection site abscesses; body areas persistently inflamed, swollen or bruised; recent surgery (within past eight weeks); severe incapacitating pain requiring immediate medical attention; hemorrhaging or active bleeding; or chronic severe high blood pressure or heart problems (defined as > 160/90). The 90-day time interval was an indicator of patient commitment to recovery and engagement in treatment, as over 34% of patients initiating care at this clinical site withdraw within the first 90 days.
All new patients received a physical exam which included chronic pain assessment conducted by a medical clinician prior to induction on methadone. Results were recorded in their electronic medical record (EMR). Between 5/1/2011 and 2/29/2012, 211 nonduplicated new patients initiated medication-assisted treatment for opioid dependence at the treatment agency. Of these, 78% completed intake (n = 165) and 65% (n = 137) remained in treatment for a minimum of 90 days. Of patients in treatment for ≥ 90 days, 80 (58%) reported chronic pain lasting at least six months of ≥ 4 on an NRS. A minimum of three attempts were made to contact all 80 patients to invite them to a study screening visit, at which time eligibility was confirmed. Out of concern that a participant might withdraw if they were not randomized to their preferred treatment regime, Baseline data were collected prior to randomization.
After participants completed the Baseline assessments, a research associate called an agency branch not affiliated with the research department and group assignments were made based on a random number generator to TAU or massage plus TAU.
Pain measures and other assessments used were designed to mirror the domains recommended by Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT)(30) for use in clinical trials of chronic pain. Instruments used to assess the following domains: 1) pain intensity by The Numeric Rating Scale (NRS)(28) – NRS measures pain on a 0 (no pain) to 10 (worst pain imaginable) scale, including pain in the last 24 hours, average daily pain, and worst pain; 2) emotional functioning through the Hospital Anxiety and Depression Scale (HADS)(31) – HADS clusters participants by anxiety and depression with scores of 0–7 (not depressed/anxious), 8–10 (suggestive), and 11+ (anxious/depressed) on a 0–21 scale; 3) improvement and satisfaction with treatment through the Patient Global Impression of Change (PGIC)(32,33) – PGIC measures patient-reported improvement or deterioration over time on a 7-point scale (very much worse to very much improved), and at Baseline visit this score is relative to how participants feel compared to the last month, and during the study each measurement is relative to the last visit; 4) symptoms and adverse events through the TimeLine Follow-back (TLFB);(34) and 5) substance use and treatment engagement and retention from data abstracted from participants’ electronic medical record. For questionnaires not in the public domain, investigators received permission for use.
Demographics, vitals, urine drug-screen results, average weekly methadone doses, and substance use management history were abstracted from the EMR. Nutritional risk assessment was determined by the screening tool, “Determine your Nutritional Health”.(35) Weight and blood pressure were collected at Baseline and Weeks 4, 8, and 12. Body Mass Index (BMI) determined from height and weight was included, based on previous reports between chronic pain and obesity (BMI ≥ 30),(36) as well as correlation between methadone treatment and weight gain.(37)
Prior to study launch, research staff and LMTs discussed whether the participants would report improvements in pain levels after massage or if they believed pain was such an integral part of their identity that to report improvement would be to lose a sense of self. Because pain is subjective and there are many ways it can be conceptualized,(38) participants were asked whether they believed they could ever be pain-free and what would it take to be pain-free. This variable is referred to as “pain-free.” Additionally, participants’ chronic pain treatment history was documented. The constructs of self-actualization and self-efficacy (i.e., a person’s belief that they can perform a behavior(39)) may influence pain management and treatment outcomes.(40) There is documentation of this construct, linking self-efficacy, coping strategies, and chronic pain management.(40) This information was collected from participants to assess if there is a relationship between ability to be pain-free/self-efficacy and changes in primary outcome measures.
Data were managed using REDCap (Research Electronic Data Capture) electronic data capture tools hosted at CODA, Inc.(41) REDCap is a secure, web-based application designed to support data capture for research studies, providing: 1) an intuitive interface for validated data entry; 2) audit trails for tracking data manipulation and export procedures; 3) automated export procedures for seamless data downloads to common statistical packages; and 4) procedures for importing data from external sources.(41)
All participants, regardless of randomization group, continued to receive their usual care including daily methadone dosing, weekly random urine drug-screen testing, at least one weekly group session, and monthly individual counseling sessions.
The massage protocol (Appendix A) was designed by an LMT instructor at a local accredited LMT school with 12 years of experience. The protocol was a tightly scripted 50-minute session, conducted once a week for the first eight weeks of the study. Individual sessions were conducted on massage tables in private rooms. The number of sessions was based on recommendations from the massage consultant and to be consistent with the literature.(21) To maintain fidelity, selected sessions were observed, timed, and reviewed by the lead LMT.
The study interventions were conducted by one of four LMTs, all of whom were licensed in the state of Oregon. All LMTs had bachelor degrees prior to attending LMT schools, expressed interest in research, had experience with marginalized populations, and were willing to be trained to and follow a standardized Swedish massage protocol. LMTs were not assigned to a specific participant. This approach was to minimize potential bias introduced by a relationship between the LMT and participant rather than from a positive massage effect. Given the number of participants who had previously exchanged sex for goods or services, male LMTs treated males and female LMTs treated females. Issues or concerns regarding gender orientation were not raised in the course of the trial.
Data were collected by trained research associates. Given the nature of the design, it was not possible to blind staff to group assignment. Data were collected prior to massage intervention, as the goal was to assess how the participants felt on average during the previous week. Data and survey instrument collection varied by the visit week (Table 1) and instrument instructions (i.e., some assessments were appropriate for weekly administration, others monthly, etc.). Visits at Baseline, Weeks 4, 8, and 12 were longer in duration. Baseline assessments were repeated at Weeks 4, 8, and 12. Progress notes completed by the research associates and LMTs after each study session provided a narrative record of the visit. All assessments were repeated at Week 12 to evaluate the durability of massage effect.
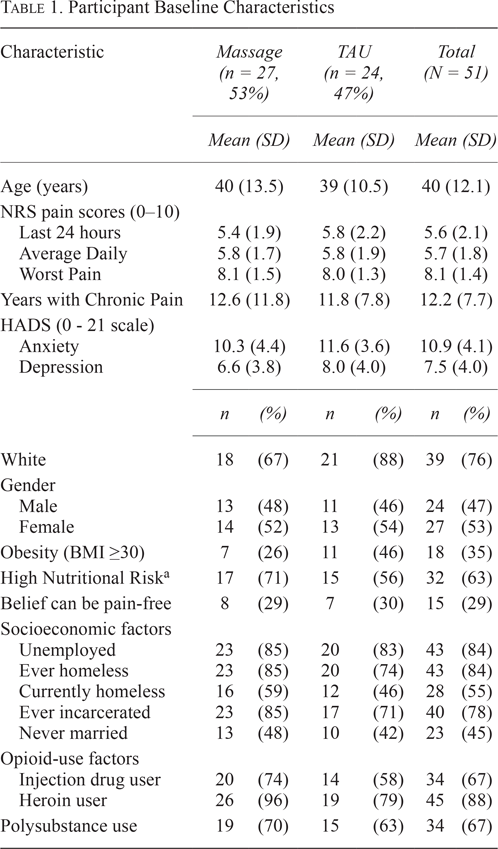
Table 1.
Participant Baseline Characteristics

All participants received a $20 gift card to a local retail supermarket chain for each completed visit. Cash incentives were not used, given the concern that cash might trigger drug-seeking behaviors.
From REDCap, data were imported into STATA 12(42) and SAS 9.2.(43) Descriptive statistics were computed for all study variables. Crude differences between massage and TAU groups were assessed using t tests for continuous measures and chi-square tests when categorical. Baseline health status measures were also compared between each group to identify any differences prior to intervention. The analysis of treatment effect on pain measured by the NRS pain scale used the data collected at Baseline, Weeks 4, 8, and 12. Clinically significant differences in NRS pain measures were based on published recommendations(44,45) and were defined a priori as a 2-point change. Comparing the percent of responders (percentages of participants that meet a threshold of percentage decrease in pain) was used to measure the differences between NRS pain scores of two groups.(46) Differences between the treatment groups’ PGIC scores were tested using the Mann-Whitney U test for Baseline and Weeks 4, 8, and 12. Given the small number of very much worse responses (n = 3) on the PGIC, this category was grouped with the much worse category.
Of the identified target population of 80 patients, 18 (23%) had exclusionary conditions, the most common of which were medical (56%) and currently pregnant (33%). Of the remaining 62 patients, 3 (5%) refused and 7 (11%) never responded to three recruitment contact efforts. We enrolled 52 (84%) of eligible patients; however, one massage participant withdrew consent prior to Week 1 because of the inconvenience of keeping a scheduled massage appointment (Appendix B).
For enrolled participants, heroin was the most-reported drug at intake (88%) and injection drug use was the primary route of use (67%). The majority of study participants were female (53%), with a mean age of 40 years, currently homeless (55%), unemployed (84%), at high nutritional risk (63%), and had been incarcerated at least once (78%). For both massage and TAU, participants’ average daily pain was 5.8 on a 0–10 NRS scale, with an average duration of 12.2 years with chronic pain. Massage and TAU groups did not differ on demographic characteristics at Baseline (Table 2).
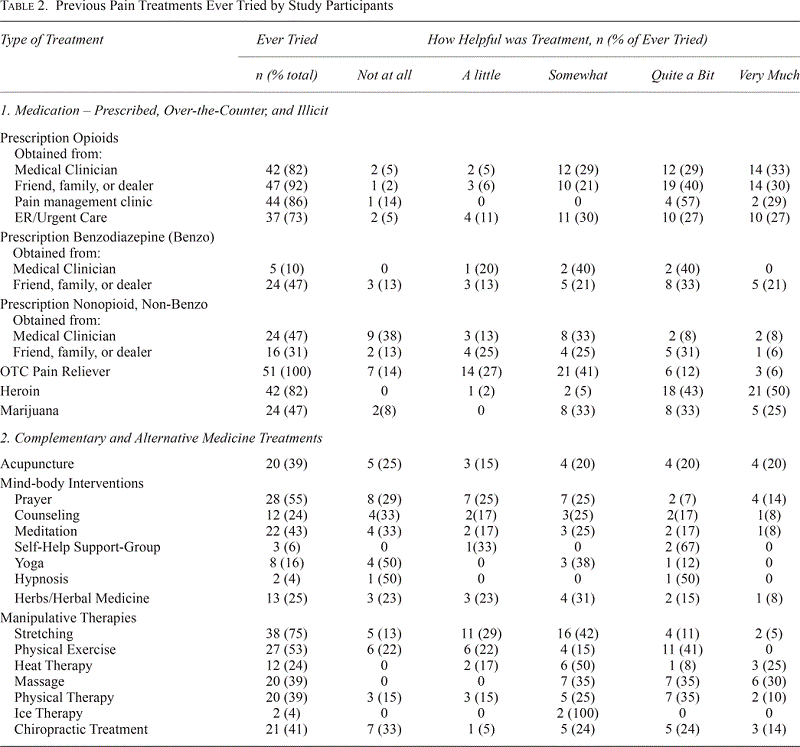
Table 2.
Previous Pain Treatments Ever Tried by Study Participants
Approximately three quarters (78%) of participants reported their chronic pain was the sequela of some type of injury sustained from: a) car/motorcycle accidents (n = 12, 23%); b) sports injuries (n = 10, 19%); c) work-related injuries (n = 9, 18%); d) falls/accidents (n = 6, 12%); or e) partner abuse (n = 3, 6%). The remaining causes were from other medical conditions (i.e., headaches, back pain (n = 6, 12%); pregnancy (n = 3, 6%); or unknown cause (n = 2, 4%)). Previous to study participation, almost all (90%) of participants felt their chronic pain had worsened over time. A majority (57%) reported weight gain since their pain began. Few participants (10%) reported seeking treatment for their pain outside of the substance-use treatment facility. A majority of participants (55%) reported that their chronic pain was part of the reason they sought methadone maintenance versus other forms of opioid-dependence treatment without analgesic properties, and 85% of heroin users believed their chronic pain contributed to their heroin use.
All participants had previously tried various remedies for managing their chronic pain, involving traditional pharmacologic, illicit, and CAM treatments with varying levels of relief (Table 3). Almost all, 92%, used illicit opioids and found them to be at least somewhat helpful in alleviating their pain. Overall, opioids, including heroin, were reported as the most helpful medication treatment. The most common complementary treatment was stretching (75%) and the majority of those found it to be at least somewhat helpful.
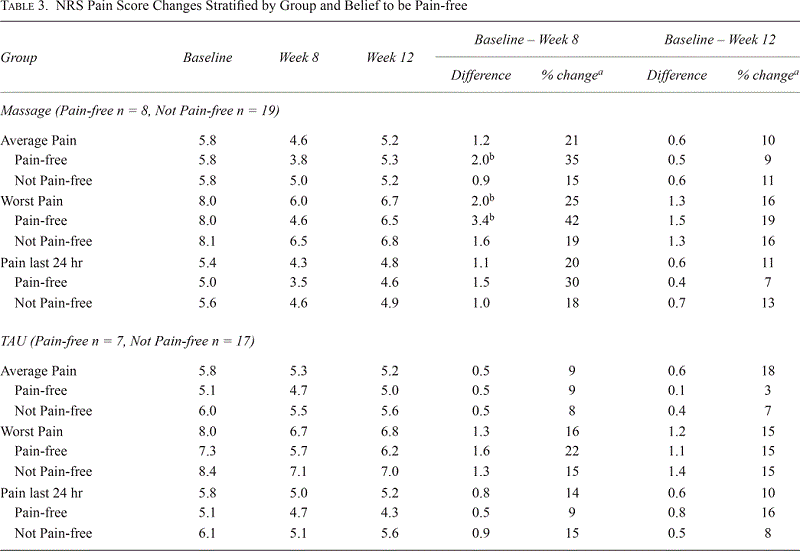
Table 3.
NRS Pain Score Changes Stratified by Group and Belief to be Pain-free
Pain scores were not associated with anatomic site on the body, type of chronic pain, or initial severity of pain. For the three NRS pain measures — pain in last 24 hours, average pain in last week, and worst pain in last week (Figure 1) — massage participants reported lower pain scores relative to the TAU group for Weeks 4 and 8 and no difference at Week 12. For both TAU and massage, the mean of their average pain scores for all three measures of pain were lower at Week 12 than at Baseline. The Weeks 4 and 8 improvements in the massage group did not attain clinical or statistical significance, except for worst pain at Week 8.
|
|
||
|
Figure 1. Mean pain scores for massage and TAU groups by week. |
||
Participants were consistently more anxious than depressed regardless of treatment group. Depression scores were in the suggestive group, 8–10, while anxiety scores reflected suggestive to full anxiety. For both depression and anxiety, TAU had slightly higher levels from Baseline through Week 12. Anxiety and depression scores remained stable and did not change throughout the trial.
The PGIC measure presented the greatest significant difference between the massage and TAU groups. The groups began the study with no change in PGIC. At both Weeks 4 and 8, massage patients reported statistically significant improvement over TAU for minimally improved and much improved, p = .005 and p < .001, respectively (Figure 3). This difference was not sustained at the Week 12 follow-up visit, p = .95, where massage participants reported higher levels of feeling minimally to much worse than TAU.
|
|
||
|
Figure 3. Patient Global Impression of Change (PGIC) by group and week. |
||
There were no adverse events related to the intervention reported during the trial.
For the biologic measures of blood pressure and urine drug screens, there were no changes observed from Baseline. Approximately 60% of participants (58.8%), consistently had urine drug screens negative for nonprescribed substances other than THC. Through Week 12, there was no difference in urine drug screen results between massage and TAU groups ( p = .95). No improvements or differences in blood pressure between massage and TAU were observed as found in a previous meta-analysis.(47) Regardless of group assignment, there were no changes in the prescribed doses of methadone. Obesity (BMI ≥ 30) was not statistically associated with pain. Weight gain of 10 or more pounds was measured in 49% of participants, regardless of group assignment.
Slightly more than a quarter of participants (30%) felt that they could ever be pain-free, which was not statistically associated with Baseline level of pain. The reported ability to be pain-free or not pain-free did not change throughout the course of the trial regardless of treatment group assignment. Participants reporting they could never be pain-free most commonly stated they would need to die, have a new body, or be rich to become pain-free. A significant decrease in NRS pain was reported in the pain-free subgroup of participants and this reached clinical significance — a change of two points — in the massage group when comparing Baseline to Week 8 (Table 3). Regardless of Baseline pain level, those who self-identified as having the potential to improve had larger decreases in pain. At Week 8 (Table 3) in the massage group, this magnitude of change for average pain was 2.0 for participants who could see themselves being pain-free compared to 0.9 who could not. This difference is even wider for worst pain, 3.4-point change from Baseline to Week 8 compared to 1.6-point change. Figure 2 shows the percent of responders in the massage group who saw themselves as being able to be pain-free were greater than all other groups for each threshold (10%, 30%, and 50%). Statistical significant treatment effect was seen in the pain free group at the 30% threshold for average pain in Week 8, z = −2.16, p = .03.
|
|
||
|
Figure 2. Percent of participants responding (percent decrease in NRS pain scores) from Baseline to Week 8. |
||
All Baseline characteristics were collected in full. There were more missing data in the TAU (6%) group than the massage (3%) group. Overall study completion rates were 85% in massage and 75% in TAU. Missing an appointment was not significantly associated with group assignment; did not follow a monotone missing pattern (i.e., once missed a visit never to return). Given evidence from recent papers on the discussion of missing data in a longitudinal study,(48,49) multiple imputation was not performed, as the percentage of missing was low.
The opioid-dependent study population was largely homeless, unemployed, and averaged 12 years with chronic pain. The participants had tried a range of traditional, illicit, and CAM treatments for pain management. Opioids, prescribed and illicit, were reported as being the most helpful in alleviating pain. The frequency of pain treatments ever tried was similar to the array reported by Barry and colleagues(50) in their study of methadone patients. For the primary outcome measure of pain intensity, the massage group reported greater improvements in pain. However, these only reached clinical significance for worst pain at Week 8. Assessing participants’ self-activation may be a valuable tool for determining readiness to accept and engage in treatment interventions. Although factors, such as self-activation, are known to moderate the relationship between beliefs and health behaviors, the mechanisms underlying these effects are unclear.(51) The subgroup self-identified as pain-free reported the greatest clinically significant improvements in all pain measures. There was no durability of treatment effect, as improvements in pain observed in the pain-free subgroup were extinguished by Week 12.
There were no changes in anxiety or depression levels for the massage and TAU groups. One likely explanation is the extreme marginalization of this patient population. A once-a-week massage treatment which has an ameliorative effect on depression in some patient populations,(22,23) may be unlikely to reduce anxiety or depression in a group of largely unemployed, marginalized patients early in substance use recovery.
Consistently, improvements in pain scores are associated with positive changes in patient’s Global Impression of Change (PGIC),(52) but this concordance was not found in this study. Overall there were no clinically significant improvements in the three NRS pain scores relative to the TAU or to Baseline measures; however, participants reported statistically significant improvements in PGIC values relative to Baseline for Weeks 4 and 8. At Week 12, the positive effect was diminished and massage participants reported the highest levels of being much worse or minimally worse. Review of participant satisfaction surveys suggests that this was due to terminating the massages which they had come to appreciate for pain management.
Overall, 49% of the study population gained 10 or more pounds in the period from initial treatment intake to the Week 12 visit, a period of approximately six months. There were no differences in BMI changes between the massage or TAU groups and BMI scores were not associated with pain scores. Given the high nutritional risk at Baseline and subsequent significant weight gain over six months, implementation of targeted nutritional counseling throughout treatment seems appropriate.
There are several important limitations to this feasibility trial, such as small sample size, absence of a true placebo, variation in types of chronic pain, and restriction to one massage type. Based on input from the study LMTs, future work should modify the Swedish massage protocol through expansion of massage scope and inclusion of other techniques. Appropriate participants may be patients with pain in a specific anatomic location (e.g., lower back) or chronic pain of a specific type (e.g., fibromyalgia). The current trial had insufficient sample size to consider anatomic location, type or cause of pain, or the role of belief in being pain-free. Potential self-activation characteristics of patients most amenable to massage intervention should be identified at intake through screening.(53,54) These data should be incorporated into future protocol development and revisions.
Findings from this trial are preliminary. Given the low percentage of participants who felt they had the potential to be pain-free, future nonpharmacologic treatments for pain in addiction patients may wish to target approaches which incorporate a patient’s activation or self-efficacy involvement in treatment. In this high-risk and low income population, massage did not contribute to overall clinically significant improved pain levels, quality of life measures, or treatment outcomes; rather, it was limited to a small, more empowered, subgroup of participants.
In this population of substance abuse patients with chronic pain, clinically significant improvement in pain was not observed in the intervention group receiving eight weekly Swedish massage sessions. However, massage participants who felt they could be pain-free reported clinically significant reductions in pain and improved treatment satisfaction. Future nonpharmacologic pain research in marginalized substance-using populations may wish to consider some of the challenges and limitations faced in this project and include examination of the role of self-efficacy/activation in patients’ beliefs of their pain. Targeted treatment plans may allow providers to focus on the role of self-efficacy, potentially resulting in better coping methods, chronic pain management, and improved treatment outcomes.
Right upper extremity: 3 min
Undrape extremity
Effleurage to entire upper extremity to spread oil (1 min)
Petrissage to upper arm followed by effleurage to entire upper extremity (1 min)
Petrissage to forearm, hand and fingers followed by effleurage to entire upper extremity (1 min)
Redrape upper extremity
Left upper extremity: 3 min
Undrape extremity
Effleurage to entire upper extremity to spread oil (1 min)
Petrissage to upper arm followed by effleurage to entire upper extremity (1 min)
Petrissage to forearm, hand, and fingers followed by effleurage to entire upper extremity (1 min)
Redrape upper extremity
Left lower extremity: 7 min
Undrape extremity
Effleurage to entire anterior, medial, and lateral lower extremity to spread oil (1 min)
Petrissage to front of thigh (1 min)
Effleurage entire anterior and lateral lower extremity and petrissage and friction around patella (30 sec)
Petrissage and friction to anterior, medial, and lateral leg (1 min 30 sec)
Effleurage entire lower extremity and petrissage and friction around ankle joint (30 sec)
Petrissage, friction and milking to foot (1 min 30 sec)
Effleurage to entire anterior, medial, and lateral lower extremity, and redrape; cupping tapotement to entire lower extremity with vibration around kneecap (1 min)
Right lower extremity: 7 min
Undrape extremity
Effleurage to entire anterior, medial and lateral lower extremity to spread oil (1 min)
Petrissage to front of thigh (1 min)
Effleurage entire anterior and lateral lower extremity and petrissage and friction around patella (30 sec)
Petrissage and friction to anterior, medial, and lateral leg (1 min 30 sec)
Effleurage entire lower extremity and petrissage and friction around ankle joint (30 sec)
Petrissage, friction and milking to foot (1 min 30 sec)
Effleurage to entire anterior, medial, and lateral lower extremity, and redrape; cupping tapotement to entire lower extremity with vibration around kneecap (1 min)
Trunk: 4 min
Circular kneading clockwise around abdomen through the drape and friction up along sternum through the drape (1 min)
Effleurage upper pectoral area to spread oil, followed by petrissage and friction to upper pectoral area (1 min)
Effleurage to posterior neck to spread oil; petris-sage and friction along left side of neck from C1 to occiput (1 min)
Petrissage and friction along right side of neck from C1 to occiput (1 min)
Turning participant: 1 min
Therapist holds top sheet while participant turns over into face-down (prone) position, with head and face in face cradle; participant has option of having pillow under the knees (1 min)
Back: 10 min
Undrape back
Effleurage to entire back from sacrum to occiput to spread oil (1 min)
Petrissage to right side of back from sacrum to shoulder joint (1 min)
Cross-fiber friction along left erector spinae muscles from sacrum to occiput (1 min)
Mobilization of scapula with friction to medial and lateral border (1 min)
Effleurage to entire back from sacrum to occiput (1 min)
Petrissage to left side of back from sacrum to shoulder joint (1 min)
Cross-fiber friction along right erector spinae muscles from sacrum to occiput (1 min)
Mobilization of scapula with friction to medial and lateral border (1 min)
Effleurage and petrissage to posterior neck (1 min)
Effleurage to entire back from sacrum to occiput (1 min); redrape back
Left lower extremity: 6 min
Undrape lower extremity
Effleurage to entire posterior, medial, and lateral lower extremity (1 min)
Petrissage to posterior, medial, and lateral thigh (1 min)
Friction to ITB (1 min)
Effleurage to entire posterior, medial, and lateral lower extremity (1 min)
Petrissage to posterior, medial, and lateral leg (1 min)
Friction around ankle and milking of foot followed by effleurage to entire posterior medial and lateral lower extremity (1 min); redrape lower extremity
Right lower extremity: 6 min
Undrape lower extremity
Effleurage to entire posterior, medial, and lateral lower extremity (1 min)
Petrissage to posterior, medial, and lateral thigh (1 min)
Friction to ITB (1 min)
Effleurage to entire posterior, medial, and lateral lower extremity (1 min)
Petrissage to posterior, medial, and lateral leg (1 min)
Friction around ankle and milking of foot followed by effleurage to entire posterior medial and lateral lower extremity (1 min); redrape lower extremity
Finishing strokes: 3 min
Soft fist beating, cupping, and hacking tapotement to entire back (1 min)
Cupping tapotement to left posterior lower extremity from buttocks to foot (1 min)
Cupping tapotement to right posterior lower extremity from buttocks to foot (1 min)
Therapist informs participant that the session is over, and instructs the participant to get off the table and get dressed after therapist leaves the room.
|
|
||
We gratefully acknowledge the backing of the Massage Therapy Foundation for funding this project. We acknowledge the help of Tee J. Ford, LMT for her development of the massage protocol, LMT training, and fidelity testing. The compassionate caring of the LMTs carried this trial with special thanks to Aaron Selmanson and Liorah Riutzel. We recognize the support of our colleagues at CODA, Inc. who made a variety of contributions to the development, implementation, and conduct of the trial: Rachael Baker, Lyndsy Martin, Kasie Cloud, and Allison O’Neill. We are grateful for the generosity and support of East West College. We acknowledge the in-kind support from the CODA, Inc. administration. Thanks to Dennis McCarty, PhD for his thoughtful edits, suggestions, and support throughout the trial. Finally, a special thanks to all of our patients and study participants who challenge us daily to improve their quality of life. The granting agency, The Massage Therapy Foundation, had no role in the study design, data collection or analyses, interpretation of the findings, in the writing of this manuscript, or in its submission for publication.
The authors declare there are no conflicts of interest. Portions of the data were presented as an oral presentation at the International Massage Therapy Research Conference, April 2013, Boston MA.
1. Johannes CB, Le TK, Zhou X, Johnston JA, Dworkin RH. The prevalence of chronic pain in United States adults: results of an Internet-based survey. J Pain. 2010;11(11):1230–1239.

2. Meldrum ML. A capsule history of pain management. JAMA. 2003;290(18):2470–2475.

3. Jamison RN, Kauffman J, Katz NP. Characteristics of metha-done maintenance patients with chronic pain. J Pain Symptom Manage. 2000;19(1):53–62.

4. Rosenblum A, Joseph H, Fong C, Kipnis S, Cleeland C, Portenoy RK. Prevalence and characteristics of chronic pain among chemically dependent patients in methadone maintenance and residential treatment facilities. JAMA. 2003;289(18):2370–2378.

5. Trafton JA, Oliva EM, Horst DA, Minkel JD, Humphreys K. Treatment needs associated with pain in substance use disorder patients: implications for concurrent treatment. Drug Alcohol Depend. 2004;73(1):23–31.
6. Peles E, Schreiber S, Gordon J, Adelson M. Significantly higher methadone dose for methadone maintenance treatment (MMT) patients with chronic pain. Pain. 2005;113(3):340–346.

7. Ilgen MA, Trafton JA, Humphreys K. Response to metha-done maintenance treatment of opiate dependent patients with and without significant pain. Drug Alcohol Depend. 2006;82(3):187–193.
8. Sheu R, Lussier D, Rosenblum A, Fong C, Portenoy J, Joseph H, et al. Prevalence and characteristics of chronic pain in patients admitted to an outpatient drug and alcohol treatment program. Pain Med. 2008;9(7):911–917.

9. Barry DT, Beitel M, Joshi D, Schottenfeld RS. Pain and substance-related pain-reduction behaviors among opioid dependent individuals seeking methadone maintenance treatment. Am J Addict. 2009;18(2):117–121.


10. Caldeiro RM, Malte CA, Calsyn DA, Baer JS, Nichol P, Kivlahan DR, et al. The association of persistent pain with out-patient addiction treatment outcomes and service utilization. Addict. 2008;103(12):1996–2005.
11. Barry DT, Beitel M, Cutter CJ, Joshi D, Falcioni J, Schottenfeld RS. Conventional and non-conventional pain treatment utilization among opioid dependent individuals with pain seeking methadone maintenance treatment: a needs assessment study. J Addict Med. 2010;4(2):81–87.
12. Potter JS, Shiffman SJ, Weiss RD. Chronic pain severity in opioid-dependent patients. Am J Drug Alcohol Abuse. 2008;34(1):101–107.
13. Potter JS, Prather K, Weiss RD. Physical pain and associated clinical characteristics in treatment-seeking patients in four substance use disorder treatment modalities. Am J Addict. 2008;17(2):121–125.

14. Dhingra L, Masson C, Perlman DC, Seewald RM, Katz J, McKnight C, et al. Epidemiology of pain among outpatients in methadone maintenance treatment programs. Drug Alcohol Depend. 2013;128(1–2):161–165.

15. Rosenblum A, Marsch LA, Joseph H, Portenoy RK. Opioids and the treatment of chronic pain: controversies, current status, and future directions. Exp Clin Psychopharm. 2008;16(5):405–416.
16. Rosenblum A, Parrino M, Schnoll SH, Fong C, Maxwell C, Cleland CM, et al. Prescription opioid abuse among enrollees into methadone maintenance treatment. Drug Alcohol Depend. 2007;90(1):64–71.

17. Wiest K, Colditz JB, Carr K, Asphaug VJ, McCarty D, Pilkonis PA. Pain and emotional distress among substance-use patients beginning treatment relative to a representative comparison group. J Addict Med. 2014;8(6):407–414.


18. Hardt J, Jacobsen C, Goldberg J, Nickel R, Buchwald D. Prevalence of chronic pain in a representative sample in the United States. Pain Med. 2008;9(7):803–812.

19. TIP 43: Medication-Assisted Treatment for Opioid Addiction in Opioid Treatment Programs. Rockville MD: SAMHSA; 2005.
20. Brands B, Blake J, Sproule B, Gourlay D, Busto U. Prescription opioid abuse in patients presenting for methadone maintenance treatment. Drug Alcohol Depend. 2004;73(2):199–207.

21. Tsao JCI. Effectiveness of massage therapy for chronic, non-malignant pain: a review. Evid-Based Compl Alt. 2007;4(2):165–179.
22. Rapaport MH, Schettler P, Bresee C. A Preliminary study of the effects of repeated massage on hypothalamic–pituitary–adrenal and immune function in healthy individuals: a study of mechanisms of action and dosage. J Alt Compl Med. 2012;18(8):789–797.
23. Rapaport MH, Schettler P, Bresee C. A preliminary study of the effects of a single session of Swedish massage on hypothalamic–pituitary–adrenal and immune function in normal individuals. J Alt Compl Med. 2010;16(10):1079–1088.
24. Bialosky JE, Bishop MD, Price DD, Robinson ME, George SZ. The mechanisms of manual therapy in the treatment of musculoskeletal pain: a comprehensive model. Manual Therapy. 2009;14(5):531–538.

25. Furlan AD, Brosseau L, Imamura M, Irvin E. Massage for low-back pain: a systematic review within the framework of the Cochrane Collaboration Back Review Group. Spine. 2002;27(17):1896–1910.

26. Manheimer E, Anderson BJ, Stein MD. Use and assessment of complementary and alternative therapies by intravenous drug users. Am J Drug Alcohol Abuse. 2003;29(2):401–413.

27. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV-TR (Text Revision), 4th ed. Washington, DC: APA; 2000.
28. Lasagna L. The clinical measurement of pain. Ann NY Acad Sci. 1960;86:28–37.

29. Oregon Health Department & US Department of Justice. Data Dictionary and Instructions for Matrix. Gov. of Oregon; 2014.
30. Dworkin RH, Turk DC, Farrar JT, Haythornthwaite JA, Jensen MP, Katz NP, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113(1):9–19.
31. Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiat Scand. 1983;67(6):361–70.

32. Guy W. ECDEU Assessment Manual for Psychopharmacology (DHEW Publication No. ADM 76–338). Washington, DC: US Dept. of Health, Education & Welfare; 1976.
33. Kamper SJ, Maher CG, Mackay G. Global rating of change scales: a review of strengths and weaknesses and considerations for design. J Manual & Manip Ther. 2009;17(3):163–170.
34. Sobell L, Sobell M. Timeline follow-back: A technique for assessing selfreported alcohol consumption. In: Litten R, Allen J, editors. Measuring Alcohol Consumption: Psychosocial and Biological Methods. New Jersey: Humana Press; 1992.
35. Posner BM, Jette AM, Smith KW, Miller DR. Nutrition and health risks in the elderly: the nutrition screening initiative. Am J Public Health. 1993;83(7):972–978.


36. Marcus DA. Obesity and the impact of chronic pain. Clin J Pain. 2004;20(3):186–191.

37. Herbert JR, Nichols SE Jr, Kabat GC. Indicators of nutritional status among clients from a New York City methadone treatment center. J Substance Abuse Treat. 1990;7(3):161–165.
38. Turk DC, Rudy TE. Towards a comprehensive assessment of chronic pain patients. Behav Res Ther. 1987;25(4):237–249.

39. Bandura A. Self-efficacy: toward a unifying theory of behavioral change. Psychol Rev. 1977;84(2):191–215.

40. Dolce JJ. Self-efficacy and disability beliefs in behavioral treatment of pain. Behav Res Ther. 1987;25(4):289–99.

41. Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JC. Research electronic data capture (REDCap) — a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381.

42. StataCorp. Stata Statistical Software, release 12. College Station, TX: StataCorp LP; 2011.
43. SAS Institute Inc. SAS 9.2 Software. Cary. NC: SAS Institute Inc., 2008.
44. Farrar JT, Portenoy RK, Berlin JA, Kinman JL, Strom BL. Defining the clinically important difference in pain outcome measures. Pain. 2000;88(3):287–294.

45. Farrar JT. Cut-points for the measurement of pain: the choice depends on what you want to study. Pain. 2010;149(2):163–164.

46. Dworkin RH, Turk DC, Wyrwich KW, Beaton D, Cleeland CS, Farrar JT, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008;9(2):105–121.
47. Moyer CA, Rounds J, Hannum JW. A meta-analysis of massage therapy research. Psychol Bull. 2004;130(1):3–18.

48. Peters SAE, Bots ML, den Ruijter HM, Palmer MK, Grobbee DE, Crouse JR, et al. Multiple imputation of missing repeated outcome measurements did not add to linear mixed-effects models. J Clin Epidemiol. 2012;65(6):686–695.

49. Twisk J, de Boer M, de Vente W, Heymans M. Multiple imputation of missing values was not necessary before performing a longitudinal mixed-model analysis. J Clin Epidemiol. 2013;66(9):1022–1028.

50. Barry DT, Beitel MB, Cutter CJ, Garnet B, Joshi D, Schottenfeld RS, et al. Allopathic, complementary, and alternative medical treatment utilization for pain among methadone-maintained patients: an exploratory study. Am J Addict. 2009;18(5):379–385.


51. Sheeran P, Gollwitzer PM, Bargh JA. Nonconscious processes and health. Health Psychol. 2013;32(5):460.
52. Farrar JT, Young Jr. JP, LaMoreaux L, Werth JL, Poole RM. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain. 2001;94(2):149–158.

53. Hibbard JH, Mahoney ER, Stockard J, Tusler M. Development and testing of a short form of the Patient Activation Measure. Health Serv Res. 2004;40(6).
54. Hibbard JH, Stockard J, Mahoney ER, Tusler M. Development of the Patient Activation Measure (PAM): conceptualizing and measuring activation in patients and consumers. Health Serv Res. 2004;39(4):1005–1026.


Published under the CreativeCommons Attribution-NonCommercial-NoDerivs 3.0 License .( Return to Text )
INTERNATIONAL JOURNAL OF THERAPEUTIC MASSAGE AND BODYWORK , VOLUME 8 , NUMBER 1 , March 2015