Sima Sadat Ghaemizade Shushtari,1 Ann Blaire Kennedy,2,3 Mina Jahangiri,4 Sharon Jackson White,5 Mojtaba Miladinia,1* Hossein Karimpourian6
1Nursing Care Research Center in Chronic Diseases, School of Nursing, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran,
2Department of Biomedical Sciences, School of Medicine Greenville, University of South Carolina, Greenville, SC, USA,
3Department of Family Medicine, Prisma Health, Greenville, SC, USA,
4Department of Biostatistics, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran,
5Emory University, Nell Hodgson Woodruff School of Nursing, Atlanta, GA, USA,
6Department of Medical Oncology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, IranBackground
Massage and acupressure are highly popular among cancer patients as non-invasive methods with different mechanisms that can target multiple coexisting symptoms. However, the comparative effectiveness between these two techniques is still unclear, particularly among patients with advanced cancer. Furthermore, it is unclear whether both approaches would have a synergistic effect if applied simultaneously. The main objective is to compare the efficacy of massage alone versus acupressure alone versus combined therapy (massage plus acupressure) for managing cancer-related symptom cluster of fatigue, pain, and sleep disturbance.
Methods
The 2-FAM-2 study is a four-arm, longitudinal, randomized trial comparing the efficacy of two complementary medicine techniques (massage alone vs. acupressure alone vs. combined therapy vs. control) for managing a fatigue–pain–sleep symptom cluster in patients with advanced cancer that will use a 2 × 2 factorial design with an equal allocation ratio. One hundred adult patients with advanced cancer who have all three symptoms of pain, fatigue, and sleep disturbance will be recruited. Four weeks of intervention and 4 weeks of follow-up with repeated measures will be part of the 8-week study period. The main outcome is the longitudinal trajectory (trend) in the intensity of the pain–sleep–fatigue symptom cluster over time, assessed via generalized estimating equations (GEE) at baseline, weekly during intervention (weeks 1–4), and at weeks 6 and 8 as follow-up. Self-reported fatigue, sleep disturbance, and pain items (0–10 scale) will be averaged to compute the symptom cluster intensity (SCI). Furthermore, a machine learning technique based on decision tree algorithms will be carried out to conduct a subgroup analysis aimed at predicting clinical outcomes for different interventions in homogeneous subgroups.
Discussion
The trial’s findings could be helpful in the development of clinical guidelines, individualization of intervention, as well as guiding clinical decisions and improving the quality of life of patients.
KEYWORDS: Complementary medicine; supportive care; symptom management; oncology; palliative medicine
Multiple interrelated symptoms that appear simultaneously are called a symptom cluster. Symptoms within a cluster create a synergistic effect on each other and may affect the patient’s quality of life.(1) Coexisting fatigue, pain, and sleep disturbance are a common symptom cluster, especially among people with advanced cancer.(2,3) The pain–sleep–fatigue symptom cluster affects more than half of patients with advanced cancer.(4) In oncology settings, managing a symptom cluster rather than single symptoms is a higher priority; hence, interventions that target multiple symptoms within a cluster are recommended.(5,6) Non-invasive and safe techniques of complementary and integrative medicine, including massage therapy and acupressure, can affect multiple coexisting symptoms in patients with cancer.(7,8)
Although massage and acupressure are tactile interventions aimed at symptom relief, they operate through partially overlapping yet predominantly distinct mechanisms. Shared effects include pain inhibition via the gate control theory, autonomic modulation through increased parasympathetic tone, and anti-inflammatory effects by reductions in pro-inflammatory cytokines.(9–13) However, their dominant pathways differ. Massage therapy predominantly acts through peripheral mechanisms, including myofascial release and enhancement of local circulation, as demonstrated by increased cutaneous temperature.(14) In contrast, acupressure predominantly exerts central neuromodulatory effects, such as meridian-specific brain activation and the endorphin-mediated Deqi response.(15,16) These physiological mechanisms activated by massage and acupressure have direct implications for pain, fatigue, and sleep disturbance. Given these complementary mechanisms, their combination may produce synergistic effects, simultaneously targeting peripheral and central pathways.
The effectiveness of both techniques on cancer-related pain, fatigue, and sleep disturbance has been shown in the literature.( 9,17–22) According to these studies, acupressure and massage therapy have shown promising effects and may be effective non-pharmacological options for managing cancer-related symptoms including fatigue, pain, and sleep disturbances. Although the American Society for Clinical Oncology’s clinical guidelines recommend massage therapy and acupressure for managing cancer-related symptoms,(23) the comparative effectiveness between these two complementary medicine techniques is unknown, particularly among patients with advanced cancer. The durability of the effect of each technique compared to each other and how effective each technique is in homogeneous subgroups of patients are still unclear. In addition, it is uncertain whether the beneficial effects of these methods may be enhanced when used concurrently. Hence, both interventions offer non-pharmacological and feasible approaches for patients with advanced cancer, and preliminary evidence supports the individual efficacy of these interventions in managing pain, fatigue, and sleep disturbances; however, their comparative or synergistic effects on the overall symptom cluster remain inconclusive. This knowledge gap is directly addressed by the present study. We hypothesize that both massage and acupressure will demonstrate superior efficacy compared to usual care in alleviating the cancer-related symptom cluster of pain, fatigue, and sleep disturbance. Furthermore, we anticipate that the combined therapy will produce synergistic effects, resulting in greater symptom relief than either intervention alone. Therefore, we designed the 2-FAM-2 trial in response to these gaps. The results of this study could be helpful in developing clinical guidelines, individualized treatment plans, and direct clinical decision-making.
On the other hand, the application of artificial intelligence and machine learning methods in recent cancer clinical trials has also been highlighted for personalized treatment.(24) Hence, the 2-FAM-2 trial aims to take steps in this direction by considering subgroup analyses based on machine learning. In addition to symptom cluster intensity (SCI), another important clinical consideration is the analgesic dose taken, which reflects real-world symptom management and functional benefit. Therefore, oral analgesic dose taken was included as a secondary outcome to capture potential changes in pharmacological pain management needs across study groups.
As described above, the objectives of the 2-FAM-2 trial are as follows:
The 2-FAM-2 factorial randomized trial protocol is in accordance with the Standard Protocol Items: Recommendations for Interventional Trial (SPIRIT) 2013 statement. The accessible protocol at https://irct.behdasht.gov.ir contains all the details from the World Health Organization trial registry dataset.
The 2-FAM-2 study is a four-arm, superiority, longitudinal, randomized controlled trial that will use a 2 × 2 factorial design with an equal allocation ratio measuring the efficacy of two complementary medicine techniques (massage alone vs. acupressure alone vs. combined therapy) versus usual care (institutional standard care) for managing cancer-related symptom cluster of fatigue, pain, and sleep disturbance in adult patients with advanced cancer. Four weeks of intervention and 4 weeks of follow-up with repeated measures will be part of the 8-week study period.
This study will be implemented in two academic sites, including a referral oncology hospital and an outpatient oncology clinic in affiliation with the Ahvaz Jundishpur University of Medical Sciences, located in southwest Iran.
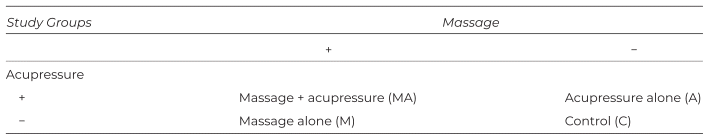
In this 2 × 2 factorial trial, the participants are assigned to one control group and three intervention groups (M: massage therapy alone; A: acupressure alone; MA: massage plus acupressure) (Table 1).
Table 1 A 2 × 2 Factorial Design for the 2-FAM-2 Trial
Arm 2—Acupressure alone: Participants will receive 30-min acupressure sessions three times a week for 4 weeks. A total of 12 acupressure sessions (360 min) will be performed for each participant. Acupressure will be performed on 15 acupoints(27): (i) GV20, (ii) PC6 (bilateral), (iii) HT7 (bilateral), (iv) LI4 (bilateral), (v) Sp6 (bilateral), (vi) St6 (bilateral), (vii) KI3 (bilateral), (viii) EX-HN3, and (ix) CV6. Table 2 shows the anatomical location of each acupoint. In the intervention, an average pressure of 3–5 kg will be applied to each point for 2 min using ventral part of the fingers (thumb/index/middle fingers) to stimulate the Deqi sensation (with the exception of the CV6 acupoint, which is stimulated with less pressure to ensure patient safety and comfort). Deqi is a special sensation in acupressure that is created by applying pressure to certain points. This sensation can include feelings such as tingling, numbness, pain, pressure, warmth, or cold. Generally, this sensation indicates successful acupressure point stimulation.(28) Prior to the intervention, participants are informed about the typical sensations associated with acupressure, which are generally brief and self-limiting.
Table 2 The Anatomical Location of the Acupressure Points
Owing to the special conditions of cancer patients, the absence of a clear massage and acupressure protocol in the oncology setting, and the emphasis of the institutional review board (IRB), oncology specialists should perform the interventions. Therefore, trained oncology nurses with backgrounds in palliative care programs will carry out the interventions. A professional massage therapist and acupuncturist will instruct the nurses until they reach an adequate level of proficiency (20 theoretical and practical sessions of 2 h each). All trained oncology nurses will conduct pilot sessions with at least five cancer patients (not enrolled in the study) under direct supervision by both the professional massage therapist and acupuncturist. To ensure precise pressure application, therapists will undergo comprehensive biofeedback training using calibrated digital algometers until they can reliably reproduce the target 3–5 kg pressure without instrumentation, as verified by blinded supervisor assessment. This training emphasizes two key competencies: (i) accurate pressure reproduction within ±0.5 kg variance when the algometer is removed and (ii) sustained maintenance of proper pressure intensity throughout the full 2-min acupoint stimulation period. Standardization will be further ensured through a three-tiered system: (i) pre-trial algometer calibration for all interventionists, (ii) mandatory monthly refresher trainings with competency re-assessment, and (iii) real-time patient-reported pressure validation using a standardized 1–10 rating scale. Any session recording pressure deviations exceeding ±0.5 kg from protocol specifications will automatically trigger therapist retraining. This multimodal approach combines objective measurement with perceptual calibration to achieve both technical precision and clinical responsiveness.
The quality and method of the interventions should be the same for all interventionists in order to prevent potential bias. All interventions will be performed in rooms with suitable environmental conditions. Separate rooms in the hospital with appropriate temperature, light, and quiet conditions will be considered.
Demographic and clinical data will be collected before randomization at baseline. In this trial, we will evaluate the patient-reported outcomes in the form of a diary. The primary outcome is the trend in the intensity of the pain–fatigue–sleep symptom cluster over time, which will be evaluated at baseline, weekly during the 4 weeks of intervention (weeks 1–4), and also in the sixth and eighth weeks as post-intervention follow-up in order to assess the lasting effect of the intervention. Participants will complete the symptom diary once a week at the end of the week (after the third intervention session). Self-reported fatigue, pain, and sleep disturbance items will be used to measure the SCI. Each symptom in the cluster will be measured using a validated 0–10 numeric rating scale (NRS), and the mean scores of the three symptoms will be computed to determine the SCI at each time point.(2,29) As a result, the possible range of SCI would be between 0 and 10. Research assistants will request patients to fill out the diary at a specific time every week, taking into account that the intensity of symptoms varies throughout the day.
Participants will be asked to rate each symptom using the following questions: (i) pain: please rate your level of pain over the past 24 h on a scale from 0 to 10, where 0 indicates “no pain at all” and 10 represents “the most severe pain imaginable”; (ii) fatigue: using a scale from 0 to 10, where 0 means “no fatigue” and 10 means “extreme fatigue,” how tired have you felt during the past 24 h?; (iii) sleep disturbance: over the past 24 h, how much have you experienced sleep problems? Please rate this on a scale from 0 to 10, with 0 meaning “no sleep disturbance” and 10 meaning “very severe sleep disturbance.” The order of symptom assessment (pain, fatigue, and sleep disturbance) on the weekly diary will remain fixed for all participants throughout the trial to ensure clarity and reduce cognitive burden.
The secondary outcome is self-reported oral analgesic dose taken, which will be evaluated at baseline, weekly during the 4 weeks of intervention (weeks 1–4), and also in the sixth and eighth weeks as post-intervention follow-up. Participants will be asked to record all analgesic medications taken during each week in the diary, including the name of the medication, dosage, and frequency.
All participants will receive phone calls from research assistants reminding them to fill out the diary and to schedule the intervention sessions. The diary will be collected on a weekly basis to input data into the statistical software. Depending on patient preference, diaries will be either collected electronically or obtained in person during scheduled clinic visits. All research assistants involved in data collection received standardized training on how to administer the diary and collect self-reported outcomes. A research assistant will document the reasons for participants withdrawing or deviating from the trial protocol. In order to avoid registration errors, the statistician will also double-check the data entry process. To ensure adherence to blinding, the study coordinator will conduct random audits to ensure that research assistants are blinded to treatment allocation. If any deviations occur, the problem will be documented and reported to the principal investigators (PIs) and data and safety monitoring committee (DSMC). The step-by-step process of 2-FAM-2 trial implementation is shown in Figure 1.
|
| ||
|
Figure 1 The step-by-step process of trial implementation. | ||
Also, participants will be monitored for potential adverse events (AEs) during each session, including bruising, discomfort, skin reaction, or any unusual responses. All AEs will be recorded by the interventionists and reported to the PIs. All interventions will be trained to monitor and report any AEs.
Participant well-being will be monitored continuously throughout the trial. During each session, trained nurses will assess for physical or emotional distress and initiate referral if needed. All participants will be reminded regularly of their right to withdraw at any time without facing any negative consequences.
Patients are recruited through the source of colleagues and posters on the sites. Recruitment of eligible patients will proceed until the target sample size is achieved. At two considered sites, two nurses will evaluate potential participants and refer those who meet the initial eligibility criteria. Afterward, trained study staff will carry out a final comprehensive evaluation to confirm eligibility. Then, written informed consent and baseline data will be obtained. The nurses who deliver the interventions are not the same nurses involved in the trial. Their only role is to perform the manual interventions according to the study protocol. This separation is considered to reduce the risk of therapeutic bias and role confusion, and ensure ethical role clarity.
Prior to enrollment, the statistician will use the blockrand package in R statistical software (R Foundation for Statistical Computing, Vienna, Austria) to create a randomization algorithm with random block sizes and an equal allocation ratio of 1:1:1:1.(30) Next, the randomization provider, who will not be involved in the trial’s screening or later phases, receives the randomization list from the statistician. The randomization provider will be blind to the study’s purposes, hypotheses, and participant characteristics. The statistician will be blinded through a coded group allocation system where study arms are labeled with non-descriptive identifiers rather than treatment names. An independent randomization coordinator will securely keep this coding key until the database is locked. During analysis, the statistician will process all outcome data using these blinded group labels. The PIs will not be aware of group allocation until the data analysis is completed. The research assistants who handle data collection alone get in touch with participants to remind them to record information in their diaries; they will be masked to group assignment. A coordinator, who is unblinded to group allocation and responsible for scheduling appointments with participants, is not involved in any other part of the trial and does not have access to the data. It is not possible to blind the participants and the interventionists, but to control the Hawthorne effect (whereby patients with higher perceived symptom scores receive more effort from the therapist), interventionists will be blinded to the pre-intervention (baseline) assessment. Interventionists will not have access to these documents. The DSMC can request to break the code for any patient, if necessary, but they are not aware of the group assignment.
The information about the participant timeline in the 2-FAM-2 trial is shown in Table 3 (SPIRIT diagram).
Table 3 SPIRIT Diagram (Standard Protocol Items: Recommendations for Interventional Trials)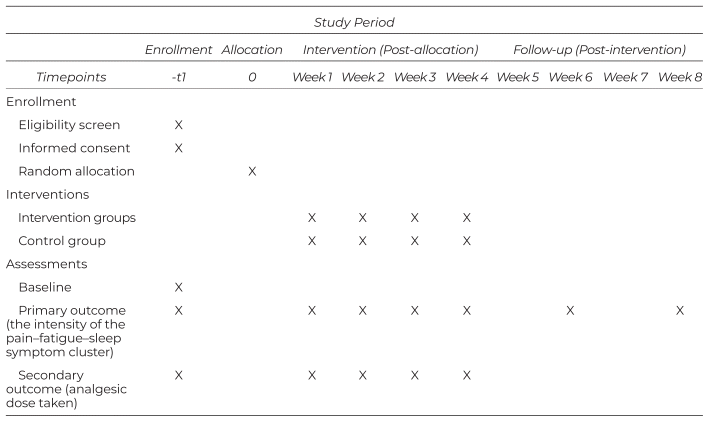
Using the WebPower package in R, a sample size of 75 participants was calculated to compare treatment groups. The parameters for this calculation included a significance level of 5%, a power of 80%, and an effect size of 0.393,(17) with four arms representing the number of groups and seven timepoints for the number of repeated measures. Additionally, a sample size of 100 subjects was determined to account for a dropout rate of 25%. Consequently, this results in 25 patients being included in each group.
This effect size (0.393) used in the sample size calculation was derived from our previous study, which evaluated the response to various massage doses for the symptom cluster of pain, fatigue, and sleep disturbances in a similar oncology population.(17) We analyzed data used by Miladinia et al. (2023) from two groups: a control group and a group receiving 3 × 30 min per week of massage. We selected the 3 × 30 min per week group because it demonstrated greater effectiveness in the aforementioned study. The partial eta squared in repeated measures analysis is defined as Based on the dataset used by Miladinia et al. (2023), the values for the “group sum of squares” and “error sum of squares” are 185.126 and 286.451, respectively. Consequently, the partial eta squared is calculated to be 0.393.
For both qualitative and quantitative variables, the descriptive statistics are presented as mean ± standard deviation and frequency (%), respectively. Analysis of variance (ANOVA) and the Kruskal–Wallis test are used to evaluate differences in quantitative variables among study arms. The difference in the frequency distribution of qualitative variables between study groups is tested using chi-square and trend chi-square tests. Normality of continuous variables is assessed using the Shapiro–Wilk test and evaluation of skewness and kurtosis. Data analysis will be conducted using the free statistical software R, and two-tailed p-values <0.05 will be considered statistically significant. Intention-to-treat analysis will be used to interpret the data. In addition, baseline variables (such as age, gender, cancer type, and stage) will be recorded and examined for potential imbalance between groups. If significant associations with outcome measures are found, these variables will be included as covariates in the statistical models to control for confounding factors.
As a superiority trial, the hypothesis is that each intervention is more effective than the control group. Between the study arms, we will compare the trend of changes in mean SCI from baseline to the eighth week. Generalized estimating equations (GEE) will be utilized to evaluate the changes over time using the package geepack in R statistical software. To assess the changes in the primary outcome variable over time, the study arms will be compared within each time point using ANOVA based on the Bonferroni correction to adjust the type I error if there is a significant interaction effect between the study arms and the time factor (from the baseline to the eighth week). If there is no normal distribution for the primary outcome variable, the changes over time will be assessed using nonparametric repeated-measure analysis in the nparLD package.(31) Furthermore, in this trial, efficacy will be defined as a ≥30% reduction in the pain–fatigue–sleep SCI at week 4 compared to the baseline, which corresponds to the established minimal clinically important difference in symptom intensity measured on a 0–10 NRS.(32)
Analgesic doses will be converted to oral morphine equivalent doses (mg/24 h) using equianalgesic dose ratios for comparisons.(33) GEE will be utilized to evaluate the changes over time using the package geepack in R statistical software. If there is no normal distribution for the secondary outcome variable, the changes over time will be assessed using nonparametric repeated-measure analysis in the nparLD package. In addition to assessing overall symptom severity, the anatomical location of pain is recorded and taken into account in secondary analyses to examine whether treatment effectiveness varies based on pain location.
The glmertree package will be utilized to conduct a linear mixed-effect model tree for predicting the efficacy of interventions in different subgroups of patients on the basis of important demographic and clinical characteristics (including age, gender, cancer type, intensity of pain–fatigue–sleep in baseline). These subgroups are defined by the tree itself, based on the splits and the treatment effects at each node. The process is that the data are sequentially divided based on variables that have a significant effect on the treatment effect. Each new division is made according to variables that make a significant difference in the treatment response. Finally, each terminal node in the tree represents a subgroup that may have a different treatment response. This method allows the clinician to identify different subgroups that respond differently to treatment and to determine a more effective treatment approach for each subgroup. This technique is a cutting-edge flexible tree approach for longitudinal data subsets.(34) Tree-based algorithms have benefits over parametric models, such as being easily interpreted, handling nonlinear relationships, and extracting homogeneous subgroups. These algorithms can identify patients needing different intervention strategies and guide clinicians in shared decision-making with patients.(35)
To alleviate missing values, we intend to use the CopyMean method to impute missing data of longitudinal quantitative response variables.(36,37) To comprehend the computational technique associated with this method, time-dependent variable is captured at t timepoints for every subject. Here, the sequence yi = (yi1, yi2,…,yit) defines a trajectory for cluster i. Let yik display a missing value at a given time-point k for cluster i. If timepoints as a < k < b exist and yia and yib are not missing, then yik is non-monotone missing. If for all timepoints h > k, yih is missing, then yik is monotone missing. The imputation of missing observations using the CopyMean approach is based on two phases. Using the last observation carried forward (LOCF) method (where yik is estimated by the last observed value of the trajectory of interest), the missing values are first imputed to provide an initial approximation of these values. Next, the population’s mean trajectory is utilized to improve the preliminary approximation from the previous phase.
This trial will not include an interim analysis.
This trial is registered in the Iranian Registry of Clinical Trials (https://irct.behdasht.gov.ir; September 9, 2024; IRCT ID: IRCT20150302021307N7). The IRB in affiliation with the Ahvaz Jundishapur University of Medical Sciences has approved the 2-FAM-2 protocol (Ref. ID: IR.AJUMS. REC.1403.323). Written informed consent will be obtained in person by trained research staff. Participants will receive clear information about the study’s aims, procedures, potential risks and benefits, and their rights, including the right to refuse or withdraw from the trial at any time without consequences to their medical care. Given the vulnerability of patients with advanced cancer, special precautions will be taken to ensure ethical participation. Prior to enrollment, trained staff will assess participants’ cognitive and emotional readiness to provide informed consent. The consent process will be conducted in person, using clear and compassionate language tailored to the individual’s condition.
The trial procedure and data will be overseen by an independent DSMC. Any changes to the protocol require approval from the IRB and the DSMC. The trial’s monitoring, coordination, and data security are under the oversight of the PIs. All participant data will be handled with strict confidentiality. Data will be stored on password-protected, encrypted institutional servers, and physical documents will be kept in locked cabinets in secure locations. Access to identifiable information will be limited to authorized research personnel only. Any possible protocol deviations must be reported by the research assistants to the PIs within 48 h. The trial must be conducted in accordance with the protocol, which is followed by trained study personnel who is routinely checked by the PIs at each study site.
The 2-FAM-2 study results will be reported in peer-reviewed journals and at scientific conferences. Also, the corresponding author will provide data and statistical codes upon reasonable request.
Symptom clusters are a common occurrence for clinicians treating patients with advanced cancer.(2) Recent research emphasizes that symptom management should shift from focusing on single symptoms to multiple symptoms. However, symptom management in this population is further complicated by the experience of multiple concurrent symptoms.(38) In clinical oncology practice, the cluster of pain, fatigue, and sleep disturbance has persisted despite all attempts to manage these symptoms. As a result, integrating complementary and integrative medicine approaches with conventional treatments has grown in significance within palliative cancer care programs.(39,40) In this regard, massage and acupressure are highly popular among cancer patients as non-invasive methods with different mechanisms that can target multiple symptoms.(7) The growing emphasis on evidence-based integrative medicine has been reflected in recent clinical practice guidelines in oncology, which support the use of massage and acupressure for symptom management in cancer care.(23,41) This study is consistent with these evolving standards, reinforcing the clinical relevance of nonpharmacological interventions as part of comprehensive supportive care. However, the comparative effectiveness between massage and acupressure is still unclear. Furthermore, it is unclear whether the two approaches would have a synergistic effect if applied simultaneously. By maintaining full dosing in the combination arm, this study allows for a direct comparison of whether the simultaneous use of both techniques produces additive or interactive improvements in symptom cluster severity. The 2-FAM-2 trial is the first 2 × 2 factorial randomized controlled trial for managing the pain–fatigue–sleep symptom cluster, comparing the efficacy of massage versus acupressure versus combined therapy versus usual care in patients with advanced cancer.
A secondary outcome of this trial is the change in oral analgesic dose taken over time. By examining this outcome, the study seeks to determine whether non-pharmacological interventions may reduce the need for pharmacological pain control. Reductions in analgesic dose taken may reflect not only improved symptom control but also potential decreases in treatment-related side effects, particularly those associated with long-term opioid use. Interpreting this outcome alongside the SCI trends will provide a more holistic view of the interventions’ clinical value.
In addition, the use of machine learning-based subgroup analysis provides a new opportunity to refine our understanding of intervention effectiveness. This approach may reveal specific patient characteristics (such as baseline symptom intensity or demographic profiles) that predict greater response to one or more treatments. Such findings could generate new hypotheses for appropriate interventions in future trials and help move toward precision supportive care in oncology. Hence, to optimize clinical decision-making, the 2-FAM-2 trial will employ a decision tree-based machine learning approach for subgroup analysis.(34) The findings of this study can be useful in developing clinical guidelines, individualizing intervention, guiding clinical decisions, and improving the quality of life of patients.
Future research should focus on the current findings by incorporating longer-term follow-up assessments to evaluate the sustained effects of massage and acupressure on symptom clusters in cancer patients. Multicenter trials across diverse clinical settings would enhance generalizability, while comparison or integration with other non-pharmacological interventions could offer broader insights. Further exploration of underlying biological mechanisms and the development of personalized intervention strategies (guided by demographic, psychological, or genetic factors) may help optimize the use of complementary therapies in oncology care.
The designed trial has some limitations: (i) in the combined therapy group, massage will be administered before acupressure. While this order is based on clinical reasoning and patient comfort, future trials should assess the effectiveness of different sequencing arrangements (e.g., acupressure followed by massage) to account for potential order effects; (ii) in the combined group, the total session duration is twice as long as in the individual intervention groups to maintain the full therapeutic dose of both interventions and eliminate the risk of subthreshold dosing in each intervention. However, this longer duration may act as a confounding factor. While this design ensures complete accuracy in dosing, future studies should explore alternative designs with identical session durations to isolate the effects of the combined interventions themselves; (iii) due to the nature of manual therapies, blinding of participants and interventionists was not possible, which may introduce expectation or performance bias; (iv) the use of a passive control group, although ethically justified, may lead to attention-related bias; (v) potential recruitment bias: although participants were recruited from two oncology sites using posters and referrals, individuals volunteering for a complementary therapies trial may have a higher baseline interest in non-pharmacological interventions. This may introduce selection bias. Furthermore, while eligibility criteria were carefully defined, the possibility of selection bias cannot be ruled out, particularly if participants were more communicative or physically able to enroll. These factors may affect the generalizability of the findings; and (vi) the inclusion and exclusion criteria used in this study were designed to ensure patient safety. However, these stringent criteria (such as excluding patients with comorbidities or bone metastases) may limit the generalizability of the findings. As a result, the findings may only be more applicable to subgroups of patients with advanced cancer who have less complex health profiles.
The recruitment of participants began in May 2025 (second version of the protocol, registered in September 2024).
The authors would like to thank the Nursing Care Research Center in Chronic Diseases affiliated with the Ahvaz Jundishapur University of Medical Sciences for supporting this investigation.
The authors declare there are no conflicts of interest.
This research is funded by a research grant from the Ahvaz Jundishapur University of Medical Sciences (grant number: NCRCCD-0312). There will be no involvement from the funder in carrying out the trial procedures.
MM, SSGS, and ABK participated in the conception of the investigation. MM, SSGS, HK, SJW, and ABK had a role in designing the trial. MM, MJ, SSGS, and ABK participated in plans for data analysis. MM, SSGS, SJW, MJ and ABK drafted the initial version of the manuscript. Revisions to the manuscript were made by all authors. The final draft of the manuscript was reviewed and approved by all authors.
The corresponding author will provide the final dataset and statistical codes upon reasonable request.
Not applicable.
1. Miaskowski C, Barsevick A, Berger A, Casagrande R, Grady PA, Jacobsen P, et al. Advancing symptom science through symptom cluster research: expert panel proceedings and recommendations. J Natl Cancer Inst. 2017;109(4):djw253.
Crossref PubMed PMC
2. Kwekkeboom KL, Tostrud L, Costanzo E, Coe CL, Serlin RC, Ward SE, et al. The role of inflammation in the pain, fatigue, and sleep disturbance symptom cluster in advanced cancer. J Pain Symptom Manage. 2018;55(5):1286–1295.
Crossref PubMed PMC
3. Miladinia M, Baraz S, Ramezani M, Malehi AS. The relationship between pain, fatigue, sleep disorders and quality of life in adult patients with acute leukaemia: during the first year after diagnosis. Eur J Cancer Care (Engl). 2018;27(1):e12762.
4. He CC, Lin DM, Liu HZ, Wang FF, Guo XF, Zhang XB, et al. Nonpharmacological interventions for management of the pain-fatigue-sleep disturbance symptom cluster in breast cancer patients: a systematic review and network meta-analysis of randomized controlled trials. J Pain Res. 2023;16:2713–2728.
Crossref PubMed PMC
5. Kwekkeboom KL. Cancer symptom cluster management. Semin Oncol Nurs. 2016;32(4):373–382.
Crossref PubMed PMC
6. Simão D, Barata PC, Alves M, Papoila AL, Oliveira S, Lawlor P. Symptom clusters in patients with advanced cancer: a prospective longitudinal cohort study to examine their stability and prognostic significance. Oncologist. 2023;29(1):e152–e163.
Crossref PubMed PMC
7. Satija A, Bhatnagar S. Complementary therapies for symptom management in cancer patients. Indian J Palliat Care. 2017;23(4):468–479.
Crossref PubMed PMC
8. Yousefi M, Reihani H, Heydari M, Nasimi Doost Azgomi R, Hashempur MH. Complementary and alternative medicine use among cancer patients in Iran: a systematic review. Prev Med Rep. 2024;39:102644.
Crossref PubMed PMC
9. Bahçecioğlu Turan G, Özer Z, Yanmış S, Aksoy A. The effects of self-acupressure on pain, fatigue, and sleep quality in colon and pancreatic cancer patients receiving chemotherapy: a randomized controlled study. Cancer Nurs. 2023;46(6):457–466.
Crossref
10. Düzel B, Çam Yanik T, Kanat C, Altun Uğraş G. The effect of acupressure on pain level and hemodynamic parameters after coronary angiography: a randomized controlled study. Front Cardiovasc Med. 2023;10:1173363.
Crossref PubMed PMC
11. Yahya FD, Ahmad M, Usman AN, Sinrang AW, Alasiry E, Bahar B. Potential combination of back massage therapy and acupressure as complementary therapy in postpartum women for the increase in the hormone oxytocin. Enferm Clín. 2020;30:570–572.
Crossref
12. Rapaport MH, Schettler P, Bresee C. A preliminary study of the effects of repeated massage on hypothalamic-pituitary-adrenal and immune function in healthy individuals: a study of mechanisms of action and dosage. J Altern Complement Med. 2012;18(8):789–797.
Crossref PubMed PMC
13. Morhenn V, Beavin LE, Zak PJ. Massage increases oxytocin and reduces adrenocorticotropin hormone in humans. Altern Ther Health Med. 2012;18(6):11–18.
PubMed
14. Diego MA, Field T. Moderate pressure massage elicits a parasympathetic nervous system response. Int J Neurosci. 2009;119(5):630–638.
Crossref PubMed
15. Si X, Xiang S, Zhang L, Li S, Zhang K, Ming D. Acupuncture with deqi modulates the hemodynamic response and functional connectivity of the prefrontal-motor cortical network. Front Neurosci. 2021;15:693623.
Crossref PubMed PMC
16. Cheng HL, Yeung WF, Wong HF, Lo HT, Molassiotis A. Self-acupressure for symptom management in cancer patients: a systematic review. J Pain Symptom Manage. 2023;66:e109–e128.
Crossref PubMed
17. Miladinia M, Jahangiri M, Kennedy AB, Fagerström C, Tuvesson H, Safavi SS, et al. Determining massage dose-response to improve cancer-related symptom cluster of pain, fatigue, and sleep disturbance: a 7-arm randomized trial in palliative cancer care. Palliat Med. 2023;37(1):108–119.
Crossref
18. Lee SH, Kim JY, Yeo S, Kim SH, Lim S. Meta-analysis of massage therapy on cancer pain. Integr Cancer Ther. 2015;14(4):297–304.
Crossref PubMed
19. Cassileth BR, Vickers AJ. Massage therapy for symptom control: outcome study at a major cancer center. J Pain Symptom Manage. 2004;28(3):244–249.
Crossref PubMed
20. Kashani F, Kashani P. The effect of massage therapy on the quality of sleep in breast cancer patients. Iran J Nurs Midwifery Res. 2014;19(2):113–118.
PubMed PMC
21. Miladinia M, Baraz S, Shariati A, Malehi AS. Effects of slow-stroke back massage on symptom cluster in adult patients with acute leukemia: supportive care in cancer nursing. Cancer Nurs. 2017;40(1):31–38.
Crossref
22. Doğan MD. The effect of acupressure on fatigue in cancer patients: a meta-analysis study. Indian J Palliat Care. 2024;30(1):10–15.
Crossref PMC
23. Mao JJ, Ismaila N, Bao T, Barton D, Ben-Arye E, Garland EL, et al. Integrative medicine for pain management in oncology: society for integrative oncology–ASCO guideline. J Clin Oncol. 2022;40(34):3998–4024.
Crossref PubMed
24. Wang J, Zeng Z, Li Z, Liu G, Zhang S, Luo C, et al. The clinical application of artificial intelligence in cancer precision treatment. J Transl Med. 2025;23(1):120.
Crossref PubMed PMC
25. Sagar SM, Dryden T, Wong RK. Massage therapy for cancer patients: a reciprocal relationship between body and mind. Curr Oncol. 2007;14(2):45–56.
Crossref PubMed PMC
26. Miladinia M, Voss JG, Molavynejad S, Malehi AS, Zarea K, Nouri EM, et al. Slow-stroke back massage compared with music therapy for leukemia-related pain and fatigue: a randomized controlled trial. JCO Oncol Pract. 2021;17(11):e1614–e1621.
Crossref PubMed
27. Tan JB, Wang T, Zhao I, Polotan MJ, Eliseeva S. An evidence-based somatic acupressure intervention protocol for managing the breast cancer fatigue-sleep disturbance-depression symptom cluster: development and validation following the Medical Research Council Framework. Int J Environ Res Public Health. 2022;19(19):11934.
Crossref PubMed PMC
28. Li MY, Kwok SWH, Tan JB, Bressington D, Liu XL, Wang T, et al. Somatic acupressure for the fatigue-sleep disturbance-depression symptom cluster in breast cancer survivors: a phase II randomized controlled trial. Eur J Oncol Nurs. 2023;66:102380.
Crossref PubMed
29. Kwekkeboom K, Zhang Y, Campbell T, Coe CL, Costanzo E, Serlin RC, et al. Randomized controlled trial of a brief cognitive-behavioral strategies intervention for the pain, fatigue, and sleep disturbance symptom cluster in advanced cancer. Psychooncology. 2018;27(12):2761–2769.
Crossref PubMed PMC
30. Snow G, Snow MG. Package ‘blockrand’. The Comprehensive R Archive Network; 2013.
31. Noguchi K, Latif M, Thangavelu K, Konietschke F, Gel YR, Brunner E. Package ‘nparLD’. 2022. https://doi.org/10.32614/CRAN.package.nparLD
32. Farrar JT, Young JP Jr, LaMoreaux L, Werth JL, Poole MR. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain. 2001;94(2):149–158.
Crossref PubMed
33. Wilkie DJ, Kampbell J, Cutshall S, Halabisky H, Harmon H, Johnson LP, et al. Effects of massage on pain intensity, analgesics and quality of life in patients with cancer pain: a pilot study of a randomized clinical trial conducted within hospice care delivery. Hosp J. 2000;15(3):31–53.
34. Fokkema M, Smits N, Zeileis A, Hothorn T, Kelderman H. Detecting treatment-subgroup interactions in clustered data with generalized linear mixed-effects model trees. Behav Res Methods. 2018;50(5):2016–2034.
Crossref
35. Loh W-Y, He X, Man M. A regression tree approach to identifying subgroups with differential treatment effects. Stat Med. 2015;34(11):1818–1833.
Crossref PubMed PMC
36. Genolini, C. Jacqmin-Gadda H. Copy mean: a new method to impute intermittent missing values in longitudinal studies. Open J Stat. 2013;3(4):26–40.
Crossref
37. Genolini C, Lacombe A, Écochard R, Subtil F. CopyMean: a new method to predict monotone missing values in longitudinal studies. Comput Methods Programs Biomed. 2016;132:29–44.
Crossref PubMed
38. Chen E, Nguyen J, Cramarossa G, Khan L, Zhang L, Tsao M, et al. Symptom clusters in patients with advanced cancer: sub-analysis of patients reporting exclusively non-zero ESAS scores. Palliat Med. 2012;26(6):826–833.
Crossref
39. Coelho A, Parola V, Cardoso D, Bravo ME, Apóstolo J. Use of non-pharmacological interventions for comforting patients in palliative care: a scoping review. JBI Database System Rev Implement Rep. 2017;15(7):1867–1904.
Crossref PubMed
40. Sattari A, Miladinia M, Alinejad Mofrad S, Akbari S, Ahmadi-Mazhin S. Nursing Perspectives on Cancer-related Symptom Management in Palliative Care: A State-of-the-Art (SotA) Review Focused on Iran. Jundishapur J Chronic Dis Care. 2025;14(4):e164702.
Crossref
41. Gowin K, Muminovic M, Zick SM, Lee RT, Lacchetti C, Mehta A. Integrative therapies in cancer care: an update on the guidelines. Am Soc Clin Oncol Educ Book. 2024;44(3):e431554.
Crossref PubMed
Corresponding author: Mojtaba Miladinia, Nursing Care Research Center in Chronic Diseases, Nursing & Midwifery School, Ahvaz Jundishapur University of Medical Sciences, Golestan Blvd., Ahvaz, Iran, E-mail: miladinia.m@ajums.ac.ir, Tel: +98-61-33110000
COPYRIGHT
Published under the CreativeCommons Attribution-NonCommercial-NoDerivs 3.0 License.
International Journal of Therapeutic Massage and Bodywork, Volume 18, Number 4, December 2025