
Pik Yu Chen, MN,1* Po Chu Ng, MSocSc,2 Ara Cheuk Yin Li, FHKCA,3 Anne Kam Hung Woo, MSc,1 Derek King Wai Yau, PhD4,5*
1Pain Management Centre, Department of Anaesthesia, Alice Ho Miu Ling Nethersole Hospital, Tai Po, New Territories, Hong Kong, China,
2Department of Occupational Therapy, Prince of Wales Hospital, Shatin, New Territories, Hong Kong, China,
3Department of Anaesthesia, Pain Medicine and Perioperative Medicine, Prince of Wales Hospital, Shatin, New Territories, Hong Kong, China,
4Department of Rehabilitation Sciences, The Hong Kong Polytechnic University, Hung Hom, Kowloon, Hong Kong, China,
5Department of Anaesthesia and Intensive Care, The Chinese University of Hong Kong, Shatin, New Territories, Hong Kong, ChinaBackground
Research indicates that chronic inflammation of the fascia and an impaired healing response contribute to central pain sensitization in fibromyalgia. The International School of Bowen Therapy (ISBT)-Bowen Therapy is a manual therapy that may stimulate the flow of blood and lymph, thus activating the body’s healing mechanisms.
Objective
The main objective of this study is to determine the effectiveness of Bowen therapy in alleviating pain, enhancing functional capability, reducing sleep and mood disturbances, and improving quality of life in patients with fibromyalgia.
Methods
Seventy-eight patients were randomly assigned to either the ISBT-Bowen Therapy (BT) (n = 40) or Control (CT) (n = 38) group in this randomized controlled trial. The BT group received eight sessions of Bowen therapy over 12 weeks, in addition to their conventional pain treatment, while the CT group did not receive Bowen therapy but continued their conventional treatment. Both groups were followed for 12 weeks after treatment. The primary outcome was pain intensity, measured by the Numeric Rating Scale. Secondary outcomes included limb endurance, activity interference, sleep disturbance, psychological distress, and quality of life.
Results
The median age of the patients was 58 (interquartile range (IQR): 50–62) years, and 68 of them (87%) were female. While there was no significant difference in pain intensity over time between the two groups, patients experienced improved endurance in the lower extremities (p < 0.001) and dominant arm (p = 0.020), reduced activity interference by pain (p = 0.005), and improved mental health-related quality of life (p = 0.002) after BT.
Conclusion
ISBT-Bowen Therapy may be effective for fibromyalgia patients in improving limb endurance, reducing activity interference, and enhancing quality of life.
KEYWORDS: Bowen therapy; fibromyalgia; pain management
Fibromyalgia is a chronic pain condition characterized by widespread pain, sleep problems, mood disturbances, and various somatic symptoms.(1) Current evidence(2–6) suggests that fibromyalgia is related to the dysfunction or dysregulation of the central nervous system, leading to a clinical phenomenon known as central sensitization. Studies have reported that muscular endurance in patients with fibromyalgia was lower compared to healthy subjects, impairing their functional capacities and motivation.(7,8) These overwhelming symptoms and dysfunctions significantly impair patients’ ability to work(9) and quality of life(10) and also affect those who are close to them.(11)
Fascia, a thin layer of connective tissue that covers every organ, muscle, bone, nerve, blood vessel, and lymphatic,(12–14) may also play a role in contributing to these symptoms. These structures are interdependent and interact with each other to influence the shape, function, and support of the entire body system.(14–16) Fascia is primarily composed of collagen(14) and is sensitive to local inflammatory responses.(17) Liptan(18) explained that inflammation of the fascia alters the release of growth hormone, secretion of cytokines, and production of collagen. The resulting overproduction of collagen may lead to the formation of adhesions.(18,19) Chronic tension in the fascia sensitizes nociceptors, causing widespread pain and impairing the fascial healing response. This leads to the dysfunction of the nervous system and contributes to the development of central sensitization in patients with fibromyalgia.(18)
The application of myofascial release techniques may help break down excessive collagen adhesions and relieve fascial tension.(18–20) When this tension is released, the flow of blood and lymph can be improved, thereby activating the body’s healing mechanisms.(21) Bowen therapy, developed by an Australian osteopath, Thomas Bowen (1916–1982), in the 1950s, is a non-invasive myofascial release technique. It involves specific sequences of gentle cross-fiber moves over muscles, tendons, ligaments, and fascia.(18,22) These moves involve applying a few grams of force to the restricted fascia layer with slow and continuous pressure, either directly or indirectly. Between the moves, there are 2-min pauses, allowing the patient’s body to respond to the stimulation. This gentle force may stimulate and improve the flow of blood and lymph, thereby activating tissue repair mechanisms(23) as the tension in the affected area is released and improved.(18,22) There were no serious adverse events associated with the safety of this technique.(23,24)
In Hong Kong, occupational therapists certified by the International School of Bowen Therapy (ISBT) use this technique in government-funded hospitals. Our recent local pilot study showed that Bowen therapy reduced pain and improved cervical range of motion, occupational performance, mood, and quality of life in patients with neck pain.(23) Many of these positive effects lasted for at least 12 weeks after completing the course of Bowen therapy. Other studies have shown that Bowen therapy provided short-term benefits for multisite chronic pain,(25) sleep,(26) developmental coordination disorder,(27) and stroke rehabilitation.(28) However, its effectiveness in patients with fibromyalgia has not been explored.
The aim of this prospective, randomized controlled trial (RCT) was to determine the effectiveness of Bowen therapy in reducing pain, enhancing functional capability, alleviating sleep disturbances, reducing psychological distress, and improving quality of life in patients with fibromyalgia.
This study was a prospective RCT. It was approved by the Joint Chinese University of Hong Kong (CUHK)—New Territories East Cluster (NTEC) Clinical Research Ethics Committee (Ref. No. 2020.357-T) and registered in the clinical trial registry (ClinicalTrials.gov PRS ID: NCT04554784; https://www.clinicaltrials.gov/study/NCT04554784?term=NCT04554784&rank=1). The study was conducted in accordance with the ethical principles of the Declaration of Helsinki.
Patients were recruited from two pain management clinics at the NTEC in Hong Kong between September 2020 and December 2023. Pain physicians screened patients who met the following criteria: (i) diagnosed fibromyalgia according to the 2010 American College of Rheumatology criteria;(1) (ii) aged between 18 and 75 years; and (iii) agreed to temporarily discontinue other alternative therapies that required direct body contact (e.g., acupuncture or massage). Conventional pain treatments were allowed.
Patients were excluded if they refused to participate or refused to temporarily discontinue alternative therapies that required direct body contact during the study period; were pregnant; had a history of major joint or limb surgery, severe psychiatric illness, known malignancy, skin disease, severe cardiovascular disease, or infectious disease; were enrolled in other studies; or were taking anticoagulants.
Detailed information about the aims and procedures of the study and the right to withdraw at any time was explained to all participants.
A research assistant, who did not participate in the study, prepared the randomization sequence and allotment, placing it in brown, opaque, and sealed envelopes. A pain nurse obtained verbal and written informed consent after a detailed explanation of the study. Recruited patients were randomly allocated to either the Bowen therapy group (BT), which received Bowen therapy in addition to their conventional pain treatment, or the control group (CT), which did not receive Bowen therapy.
A designated pain nurse and an occupational therapist, both blinded to the group allocation, performed all outcome assessments. Unblinded occupational therapists and pain physicians provided Bowen therapy and conventional treatment, respectively, according to the allocation.
In the BT group, certified occupational therapists conducted all Bowen therapy sessions. To minimize the effect of other forms of alternative therapy and their influence on the effects of Bowen therapy, Bowen therapy was started at least 1 week after the screening visit, allowing a washout period after discontinuing other alternative therapies.
Bowen therapy sessions were conducted once weekly for the first four sessions and bi-weekly for the following four sessions. A pre-designed fibromyalgia pain protocol was progressively implemented over the eight sessions. The first two sessions included Bowen Therapy sequences 1, 4, 2, and 3 moves to release fascial tension in the lower and upper back and neck. “Kidney” and “Asthma” sequences of Bowen moves were added in the third and fourth sessions to resolve tension, improve fatigue, and restore the body’s natural healing process. From the fifth session onward, treatment was tailored to individual needs, with reinforcement and additional moves performed in the last two sessions (Table 1).
Table 1 Sequences of Bowen Therapy in Different Sessions
Each session lasted 20–45 min. Two-minute pauses were given between moves or when the patient appeared over-stimulated, allowing the body to respond to the stimulation. Aftercare and self-management techniques were reinforced at the end of each session to maintain proper body postures and habits, such as avoiding strenuous activities within 48 h after the procedure, encouraging daily walking for 15–20 min on level ground, and drinking plenty of water to enhance hydration and circulation, thereby activating the body’s healing mechanisms. Self-management techniques, including arm swings and shoulder exercises, were taught to enhance recovery and promote mobility. Patients were reminded to avoid sitting with crossed legs and to prevent from remaining in the same posture for prolonged periods.
In the CT group, patients did not receive Bowen therapy. The same health education pamphlet given to the BT group was also provided to the CT group at the start of the study. Although they were advised not to receive other alternative therapies apart from conventional treatment, there were no strict restrictions. Any alternative therapy received during the study period was documented.
The primary outcome was pain intensity, measured by the Numeric Rating Scale. Secondary outcomes included the Endurance Strength Test (EST)(7) for upper and lower extremities, measured by the total number of correct executions; Brief Pain Inventory (BPI)(29) for assessing pain interference with activities; Jenkins Sleep Evaluation Questionnaire (JSEQ)(30) for evaluating sleep disturbances; Hospital Anxiety and Depression Scale (HADS)(31) for measuring psychological distress and mood disturbance; and 36-Item Short-Form Survey (SF-36)(32,33) for assessing physical, mental, and functional wellness. Patients were given validated Chinese versions of these questionnaires for each assessment. Both groups were assessed at three different time points: before the treatment (baseline), at the end of the 12-week treatment (post-Rx), and 12 weeks after treatment (post-Rx 12W).
Based on our recent publication on Bowen therapy for neck pain,(23) which had a similar study design and used pain intensity as the primary outcome, the effect size was 0.69, indicating clinical significance. Assuming a similar moderate effect size (0.69) for this study, a sample size of 34 participants in each group would provide 80% power with a two-sided significance level of 0.050 to detect a difference. The total sample size required was 80, anticipating a 20% dropout rate.
Validation rules were established to avoid errors during data entry. Data cleaning was performed to identify and correct abnormal values by referring to the original records if discrepancies were found. Demographic data were presented descriptively as mean (standard deviation (SD)) or median (interquartile range (IQR)) for continuous data, depending on data normality, or as number (percentage) for categorical data. The Shapiro–Wilk test was used to test data normality. An intention-to-treat analysis was used. Generalized estimating equation models with a Gaussian distribution, identity-link function, exchangeable correlation, and robust variance were used to compare the mean differences (MDs) in outcomes between groups after adjusting for clinically relevant covariates, such as age, gender, duration of pain, number of painful areas (i.e., Widespread Pain Index (WPI) score), and any baseline variables significantly different between groups (i.e., number of specialties seen). All statistical analyses were conducted using SPSS 27.0 for Windows (SPSS Inc., Chicago, IL) and STATA 14.2 (StataCorp, College Station, TX). The level of significance was set at p < 0.050 without adjusting for multiplicity.
Eighty-eight patients were screened between September 15, 2020, and December 22, 2023 (Figure 1). Of these, eight patients were excluded (five did not meet the inclusion criteria, and three refused to participate). A total of 80 participants were randomized, with 40 evenly allocated to each group (Figure 1).
|
| ||
|
Figure 1 CONSORT flow diagram. | ||
In the CT group, one participant withdrew after the initial assessment, and one was found not to meet the inclusion criteria by the end of the study. Fifteen patients (37.5%) received other alternative therapies (acupuncture: 15) during the study period. In the BT group, all participants completed the treatment, and one of them (2.5%) received other alternative therapies (acupuncture: 1) during the study period. Seventy-eight patients were included in the intention-to-treat analysis (BT group, n = 40; CT group, n = 38). The baseline characteristics of the participants in the two groups are shown in Table 2. No adverse events related to Bowen therapy were reported.
Table 2 Demographic and Baseline Data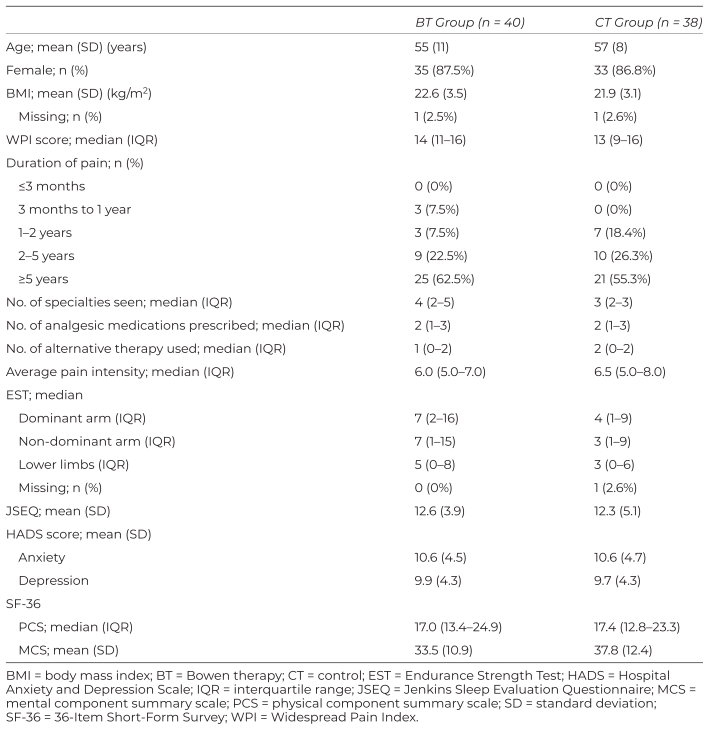
The median (IQR) averaged pain intensity reported was 6.0 (5.0–7.3), 6.0 (4.0–7.0), and 6.0 (5.0–7.0) in 78 participants at baseline, upon completion, and 12 weeks after the completion of the 12-week intervention series, respectively.
Although there was no group*time interaction effect (p = 0.519) or group effect (p = 0.595), there was a time effect (p = 0.005) on pain intensity after adjusting for age, gender, duration of pain, WPI score, and the number of specialties seen.
Upon completion of the intervention series, the BT group showed a mild reduction in pain intensity compared to baseline (MD (95% confidence interval [CI]): −0.7 (−1.3 to −0.2); p = 0.005), while the CT group did not (MD (95% CI): −0.4 (−0.8 to 0.1); p = 0.127) (Figure 2). At 12 weeks after the intervention series, no significant changes in pain intensity compared to baseline were noted in both groups (both p > 0.050).
|
| ||
|
Figure 2 Changes in the averaged pain intensity. CI = confidence interval; EST = Endurance Strength Test; Post-Rx 12W = 12 weeks post-intervention. | ||
Overall, there were differences in the changes in the endurance of lower limbs (Figure 3) and the dominant arm (Figure 4) over time (group*time p < 0.001 and p = 0.020, respectively).
|
| ||
|
Figure 3 Changes in the lower limbs endurance. CI = confidence interval; Post-Rx 12W = 12 weeks post-intervention. | ||
|
| ||
|
Figure 4 Changes in the dominant arm endurance. CI = confidence interval; EST = Endurance Strength Test; Post-Rx 12W = 12 weeks post-intervention. | ||
Significant differences were noted in the lower limbs and dominant arm at both time points (Post-Rx: p = 0.030 and p = 0.001, respectively; Post-Rx 12W: p = 0.002 and p = 0.001, respectively) when comparing with the CT group (Table 3). For the non-dominant arm, there was no group*time interaction effect (p = 0.198); however, there were group (p = 0.007) and time (p = 0.013) effects (Table 3). Significant differences were noted upon (p = 0.010) and 12 weeks (p = 0.003) after the completion of interventions when comparing with the CT group (Table 3).
Table 3 Estimates of Within-Group and Between-Group Differences for Secondary Outcome Measures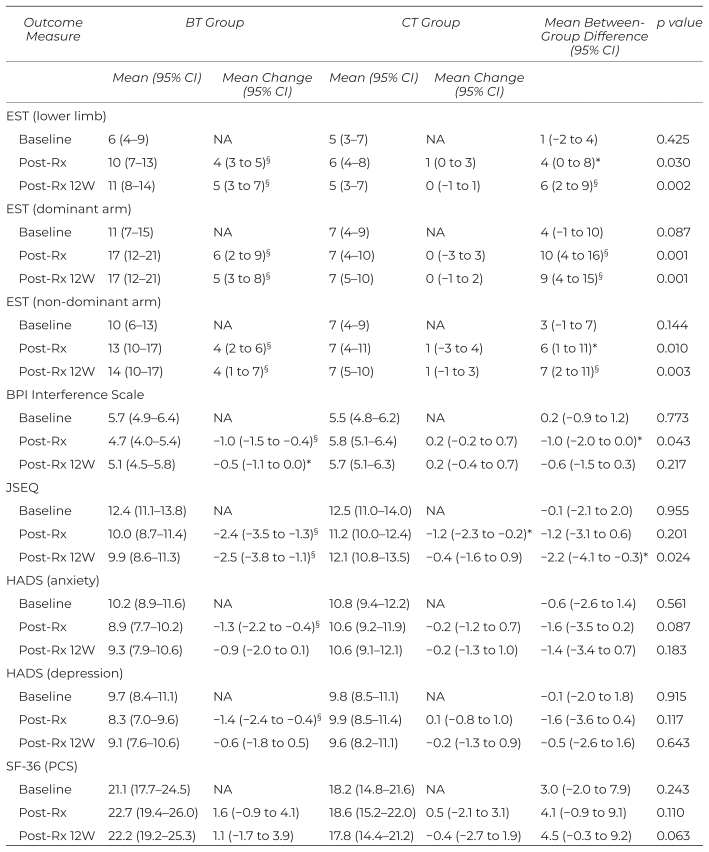
After adjusting for age, gender, duration of pain, WPI score, and the number of specialties seen, it was observed that only patients in the BT group performed more stands and curls (for both dominant and non-dominant arms) during EST upon and 12 weeks after completion of Bowen therapy (Table 3), compared to baseline levels (all p < 0.001).
After adjusting for age, gender, duration of pain, WPI score, and the number of specialties seen, a group*time interaction effect was noted on the BPI interference scale (p = 0.005) (Figure 5).
|
| ||
|
Figure 5 Changes in the Brief Pain Inventory (BPI) interference score. CI = confidence interval; Post-Rx 12W = 12 weeks post-intervention. | ||
Upon completing the interventions, the BT group had a lower BPI interference score than the CT group (p = 0.043) (Table 3).
Compared to baseline, the BPI interference scale of the BT group was reduced immediately (p = 0.001) and 12 weeks (p = 0.046) after completing Bowen therapy, but not in the CT group (Table 3).
Changes in JSEQ, adjusted for age, gender, duration of pain, WPI score, and the number of specialties seen, are shown in Table 3.
Although there was no group*time interaction (p = 0.079) or group effect (p = 0.181), there was a time effect for the JSEQ (p < 0.001). The BT group showed a significant reduction in JSEQ 12 weeks after the intervention when compared to the CT group (p = 0.024) (Table 3).
Both the BT (p < 0.001) and CT groups (p = 0.023) showed reductions in JSEQ after completing the interventions (Table 3). However, only the BT group showed a reduction in JSEQ 12 weeks after the intervention compared to baseline (MD (95% CI): −2.5 (−3.8 to −1.1); p < 0.001).
After adjusting for age, gender, duration of pain, WPI score, and the number of specialties seen, there was no group*time interaction (p = 0.277), group effect (p = 0.187), or time effect (p = 0.058). Similar results were found for depression scores; no group*time interaction (p = 0.066), group (p = 0.431), or time effect (p = 0.158).
BT group reported a mild reduction in anxiety (p = 0.003) and depression levels (p = 0.007) upon the completion of Bowen therapy compared to baseline (Table 3).
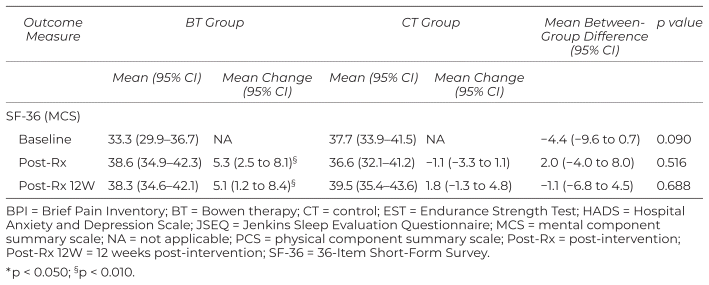
No difference in the SF-36 physical component summary score between groups was found (Table 3). After adjusting for age, gender, duration of pain, WPI score, and the number of specialties seen, there was no group*time interaction (p = 0.696), group effect (p = 0.092), or time effect (p = 0.516). However, there was a group*time interaction effect (p = 0.002) on the SF-36 mental component summary (MCS) score after adjusting for age, gender, duration of pain, WPI score, and the number of specialties seen.
The BT group reported improved SF-36 MCS at the completion of Bowen therapy (p < 0.001) and 12 weeks later (p = 0.003) compared to baseline (Figure 6), while the CT group did not show any difference over time (Table 3).
|
| ||
|
Figure 6 Changes in the SF-36 mental component summary (MCS) score. CI = confidence interval; Post-Rx 12W = 12 weeks post-intervention. | ||
The results of this study showed significant functional improvement in limb endurance strength, activity interference by pain, and mental health-related quality of life in the BT group compared to the control group, despite there was no clinically significant improvement in pain reduction and mood disturbance between the two groups.
As mentioned, the Bowen moves may stimulate and improve the flow of blood and lymph, thereby activating tissue repair mechanisms.(23) This effect is hypothesized to occur as a result of the tension in the affected area being released and circulation improved,(18,22) which can lead to overall improvements in the myofascial system. Our observed improvement in limb endurance strength and physical functioning enhances patients’ motivation to engage in more activities, thereby improving their mental health-related quality of life. This likely reflects the direct effect of Bowen therapy. On the other hand, fibromyalgia is a syndrome characterized by widespread pain, diffused sensitization, and somatic symptoms. It involves complex pathophysiological processes, including neurochemical imbalance and dysfunction in both excitatory and inhibitory mechanisms, likely mediated by multiple systems.(11) Moreover, studies revealed that patients with chronic pain demonstrated a biased interpretation of ambiguous information, favoring pain-/illness-related interpretations.(34,35) This may explain why there was no clinically significant improvement in pain reduction despite after Bowen therapy.
Most patients with fibromyalgia have lived with pain for many years and may have developed maladaptive illness behaviors and inappropriate coping skills, making them more resistant to treatment.(36) In addition, underlying bio-psycho-social stressors are known contributors to low mood and higher reported pain intensity.(37) All these factors affect pain modulation and contribute to an emotional burden that could be benefited by combining Bowen therapy with psychosocial therapy.
For managing patients with chronic pain conditions like fibromyalgia, there is no single treatment modality that can resolve all problems faced by these patients. It should focus on the improvement of functional capacity and quality of life.(38) Our result showed that even with similar levels of pain, patients could boost their functional capacity and self-help ability after receiving Bowen therapy.
We suggest combining Bowen therapy with conventional multidisciplinary chronic pain management, including but not limited to cognitive behavioral therapy and exercise, which may prolong the treatment effect and ameliorate patients’ bio-psycho-social issues. This approach may help them regain control over their lives.
There are limitations in this study. First, no sham treatment was arranged for patients in the CT group as taking part in sham treatment would have incurred costs to the patients, which would discourage them from enrolling in the study. Second, although we discouraged the participants from receiving other forms of alternative therapy with direct body contact during the study period, 1 (2.5%) and 15 (37.5%) of the patients in the BT and CT groups, respectively, did not discontinue other forms of alternative therapy, making it hard to determine the efficacy of Bowen therapy.
The benefits of ISBT-Bowen Therapy for patients suffering from fibromyalgia may include improved limb endurance strength, reduced activity interference, and enhanced quality of life. Future studies may investigate the benefits and efficacy of combining ISBT-Bowen Therapy with other treatment modalities, such as psychological therapy and physical therapy in patients with fibromyalgia.
The authors declare there are no conflicts of interest.
No sources of funding were used in this study.
PYC, ACYL, PCN, and AKHW conceived and planned the work. DKWY and PYC performed data analysis. PYC, DKWY, ACYL, and PCN wrote the paper or reviewed successive versions and took part in the revision process. All authors approved the final version.
1. Wolfe F, Clauw DJ, Fitzcharles MA, Goldenberg DL, Katz RS, Mease P, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken). 2010;62(5):600–610. https://doi.org/10.1002/acr.20140
Crossref PubMed
2. Woolf CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain. 2011;152(3 Suppl):S2–S15. https://doi.org/10.1016/j.pain.2010.09.030
Crossref
3. Rehm S, Sachau J, Hellriegel J, Forstenpointner J, Børsting Jacobsen H, Harten P, et al. Pain matters for central sensitization: sensory and psychological parameters in patients with fibromyalgia syndrome. Pain Rep. 2021;6(1):e901. https://doi.org/10.1097/PR9.0000000000000901
Crossref PubMed PMC
4. Nijs J, George SZ, Clauw DJ, Fernández-de-Las-Peñas C, Kosek E, Ickmans K, et al. Central sensitisation in chronic pain conditions: latest discoveries and their potential for precision medicine. Lancet Rheumatol. 2021;3(5):e383–e392. https://doi.org/10.1016/S2665-9913(21)00032-1
Crossref PubMed
5. Cagnie B, Coppieters I, Denecker S, Six J, Danneels L, Meeus M. Central sensitization in fibromyalgia? A systematic review on structural and functional brain MRI. Semin Arthritis Rheum. 2014;44(1):68–75. https://doi.org/10.1016/j.semarthrit.2014.01.001
Crossref PubMed
6. Cardinal TM, Antunes LC, Brietzke AP, Parizotti CS, Carvalho F, De Souza A, et al. Differential neuroplastic changes in fibromyalgia and depression indexed by up-regulation of motor cortex inhibition and disinhibition of the descending pain system: an exploratory study. Front Hum Neurosci. 2019;13:138. https://doi.org/10.3389/fnhum.2019.00138
Crossref PubMed PMC
7. Munguía-Izquierdo D, Legaz-Arrese A. Reliability and validity of a low load endurance strength test for upper and lower extremities in patients with fibromyalgia. Arch Phys Med Rehabil. 2012;93(11):2035–2041. https://doi.org/10.1016/j.apmr.2012.02.007
Crossref PubMed
8. Góes SM, Leite N, Shay BL, Homann D, Stefanello JM, Rodacki AL. Functional capacity, muscle strength and falls in women with fibromyalgia. Clin Biomech (Bristol). 2012;27(6):578–583. https://doi.org/10.1016/j.clinbiomech.2011.12.009
Crossref PubMed
9. Guymer EK, Littlejohn GO, Brand CK, Kwiatek RA. Fibromyalgia onset has a high impact on work ability in Australians. Intern Med J. 2016;46(9):1069–1074. https://doi.org/10.1111/imj.13135
Crossref PubMed
10. Hoffman DL, Dukes EM. The health status burden of people with fibromyalgia: a review of studies that assessed health status with the SF-36 or the SF-12. Int J Clin Pract. 2008;62(1):115–126. https://doi.org/10.1111/j.1742-1241.2007.01638.x
Crossref
11. Galvez-Sánchez CM, Duschek S, Reyes Del Paso GA. Psychological impact of fibromyalgia: current perspectives. Psychol Res Behav Manag. 2019;12:117–127. https://doi.org/10.2147/PRBM.S178240
Crossref PubMed PMC
12. Wilke J, Schleip R, Yucesoy CA, Banzer W. Not merely a protective packing organ? A review of fascia and its force transmission capacity. J Appl Physiol (1985). 2018;124(1):234–244. https://doi.org/10.1152/japplphysiol.00565.2017
Crossref
13. Adstrum S, Hedley G, Schleip R, Stecco C, Yucesoy CA. Defining the fascial system. J Bodyw Mov Ther. 2017;21(1):173–177. https://doi.org/10.1016/j.jbmt.2016.11.003
Crossref PubMed
14. Kumka M, Bonar J. Fascia: a morphological description and classification system based on a literature review. J Can Chiropr Assoc. 2012;56(3):179–191.
PubMed PMC
15. Bordoni B, Marelli F, Morabito B, Sacconi B. The indeterminable resilience of the fascial system. J Integr Med. 2017;15(5):337–343. https://doi.org/10.1016/S2095-4964(17)60351-0
Crossref PubMed
16. Bordoni B, Mahabadi N, Varacallo M. Anatomy, fascia. In: StatPearls. StatPearls Publishing; July 17, 2023.
17. Kondrup F, Gaudreault N, Venne G. The deep fascia and its role in chronic pain and pathological conditions: a review. Clin Anat. 2022;35(5):649–659. https://doi.org/10.1002/ca.23882
Crossref PubMed
18. Liptan GL. Fascia: a missing link in our understanding of the pathology of fibromyalgia. J Bodyw Mov Ther. 2010;14(1):3–12. https://doi.org/10.1016/j.jbmt.2009.08.003
Crossref
19. Castro-Sánchez AM, Matarán-Peñarrocha GA, Arroyo-Morales M, Saavedra-Hernández M, Fernández-Sola C, Moreno-Lorenzo C. Effects of myofascial release techniques on pain, physical function, and postural stability in patients with f ibromyalgia: a randomized controlled trial. Clin Rehabil. 2011;25(9):800–813. https://doi.org/10.1177/0269215511399476
Crossref PubMed
20. Chaitow L. Fascial well-being: mechanotransduction in manual and movement therapies. J Bodyw Mov Ther. 2018;22(2):235–236. https://doi.org/10.1016/j.jbmt.2017.11.011
Crossref PubMed
21. Ajimsha MS, Al-Mudahka NR, Al-Madzhar JA. Effectiveness of myofascial release: systematic review of randomized controlled trials. J Bodyw Mov Ther. 2015;19(1):102–112. https://doi.org/10.1016/j.jbmt.2014.06.001
Crossref PubMed
22. Wilks J, Knight I. Using the Bowen Technique to Address Complex and Common Conditions. Singing Dragon; 2014. Available from: https://books.google.com.hk/books?hl=zh-TW&lr=&id=PxE4BAAAQBAJ&oi=fnd&pg=PP2&dq=bowen+therapy+for+fibromyalgia&ots=kJUbUAP2_3&sig=JGVvbA401Sk6z8y4NHjASFdrVyk&redir_esc=y#v=onepage&q=bowen%20therapy%20for%20fibromyalgia&f=false. Accessed July 29, 2024.
23. Ying CLA, Lee TMA, Chen PY, Samy W, Au LYF, Siu CHD. The effect of ISBT-Bowen therapy in the treatment of myofascial neck pain-a randomized, single-blinded clinical trial. Int J Ther Massage Bodywork. 2023;16(2):29–38. https://doi.org/10.3822/ijtmb.v16i2.801
Crossref PubMed PMC
24. Avichal Ughreja R, Venkatesan P, Balebail Gopalakrishna D, Preet Singh Y, Vani Lakshmi R. Effectiveness of craniosacral therapy, Bowen therapy, static touch and standard exercise program on sleep quality in fibromyalgia syndrome: a randomized controlled trial. J Integr Med. 2024;22(4):473–483. https://doi.org/10.1016/j.joim.2024.06.003
Crossref PubMed
25. Lee K, Lewis GN. Short term relief of multisite chronicpain with Bowen therapy: a doubleblind, randomized controlled trial. J Bodyw Mov Ther. 2020;24(4):271–279. https://doi.org/10.1016/j.jbmt.2020.06.025
Crossref PubMed
26. Avichal Ughreja R, Venkatesan P, Balebail Gopalakrishna D, Preet Singh Y, Vani Lakshmi R. Effectiveness of craniosacral therapy, Bowen therapy, static touch and standard exercise program on sleep quality in fibromyalgia syndrome: a randomized controlled trial. J Integr Med. 2024;22(4):473–483. https://doi.org/10.1016/j.joim.2024.06.003
Crossref PubMed
27. Morgan-Jones M, Knott F, Wilcox H, Ashwin C. A pilot study of fascia Bowen therapy for 8–11 year-old boys with developmental coordination disorder. J Bodyw Mov Ther. 2019;23(3):568–574. https://doi.org/10.1016/j.jbmt.2019.02.022
Crossref PubMed
28. Duncan B, McHugh P, Houghton F, Wilson C. Improved motor function with Bowen therapy for rehabilitation in chronic stroke: a pilot study. J Prim Health Care. 2011;3(1):53–57.
Crossref PubMed
29. Keller S, Bann CM, Dodd SL, Schein J, Mendoza TR, Cleeland CS. Validity of the brief pain inventory for use in documenting the outcomes of patients with noncancer pain. Clin J Pain. 2004;20(5):309–318. https://doi.org/10.1097/00002508-200409000-00005
Crossref PubMed
30. Jenkins CD, Stanton BA, Niemcryk SJ, Rose RM. A scale for the estimation of sleep problems in clinical research. J Clin Epidemiol. 1988;41(4):313–321. https://doi.org/10.1016/0895-4356(88)90138-2
Crossref PubMed
31. Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361–370. https://doi.org/10.1111/j.1600-0447.1983.tb09716.x
Crossref PubMed
32. Ware JE Jr. SF-36 health survey update. Spine (Phila Pa 1976). 2000;25(24):3130–3139. https://doi.org/10.1097/00007632-200012150-00008
Crossref PubMed
33. Lam CL, Tse EY, Gandek B, Fong DY. The SF-36 summary scales were valid, reliable, and equivalent in a Chinese population. J Clin Epidemiol. 2005;58(8):815–822. https://doi.org/10.1016/j.jclinepi.2004.12.008
Crossref PubMed
34. Schoth DE, Liossi C. Biased interpretation of ambiguous information in patients with chronic pain: a systematic review and meta-analysis of current studies. Health Psychology. 2016;35(9):944–956. https://doi.org/10.1037/hea0000342
Crossref PubMed
35. Todd J, van Ryckeghem DML, Sharpe L, Crombez G. Attentional bias to pain-related information: a meta-analysis of dot-probe studies. Health Psychol Review. 2018;12(4):419–436. https://doi.org/10.1080/17437199.2018.1521729
Crossref
36. Borisovskaya A, Chmelik E, Karnik A. Exercise and chronic pain. In: Xiao J, ed. Physical Exercise for Human Health: Advances in Experimental Medicine and Biology, Vol. 1228. Springer; 2020. https://doi.org/10.1007/978-981-15-1792-1_16
Crossref
37. Mills SEE, Nicolson KP, Smith BH. Chronic pain: a review of its epidemiology and associated factors in population-based studies. Br J Anaesth. 2019;123(2):e273–e283. https://doi.org/10.1016/j.bja.2019.03.023
Crossref PubMed PMC
38. Nicholas M. Manage Your Pain 3rd Edition. Harper-Collins Australia; 2011. Available from: https://books.google.com.hk/books?id=B-lvlDY-QE0C. Accessed July 29, 2024.
Corresponding authors: Pik Yu Chen, Pain Management Centre, Department of Anaesthesia, Alice Ho Miu Ling Nethersole Hospital, 11 Chuen On Road, Tai Po, New Territories, Hong Kong, China, E-mail: pro910@cuhk.edu.hk, Tel: +852 55696172. Derek King Wai Yau, Department of Rehabilitation Sciences, The Hong Kong Polytechnic University, Hung Hom, Kowloon, Hong Kong, China, E-mail: derek-kw.yau@polyu.edu.hk, Tel: +852 27665396
COPYRIGHT
Published under the CreativeCommons Attribution-NonCommercial-NoDerivs 3.0 License.
International Journal of Therapeutic Massage and Bodywork, Volume 18, Number 4, December 2025